Scalp angiosarcoma: Discussion of a management plan
Images
CASE SUMMARY
A 60-year-old male presented with a 3-month history of a lump on the left vertex of his scalp. On examination, the lesion appeared erythematous with a central clearing. Initial biopsy demonstrated epithelioid angiosarcoma. No other skip lesions were noted during a mapping procedure with 8-punch biopsies. Wide local excision with a sentinel lymph-node biopsy demonstrated extensive dermal and subcutaneous fat infiltration from angiosarcoma extending to the deep aspect of the right margin, and a distinct soft-tissue deposit with a focus of angiosarcoma without any obvious association with lymph nodal tissue within the sentinel biopsy specimen. Re-excision, with accompanying split thickness skin grafting, was performed to obtain negative margins.
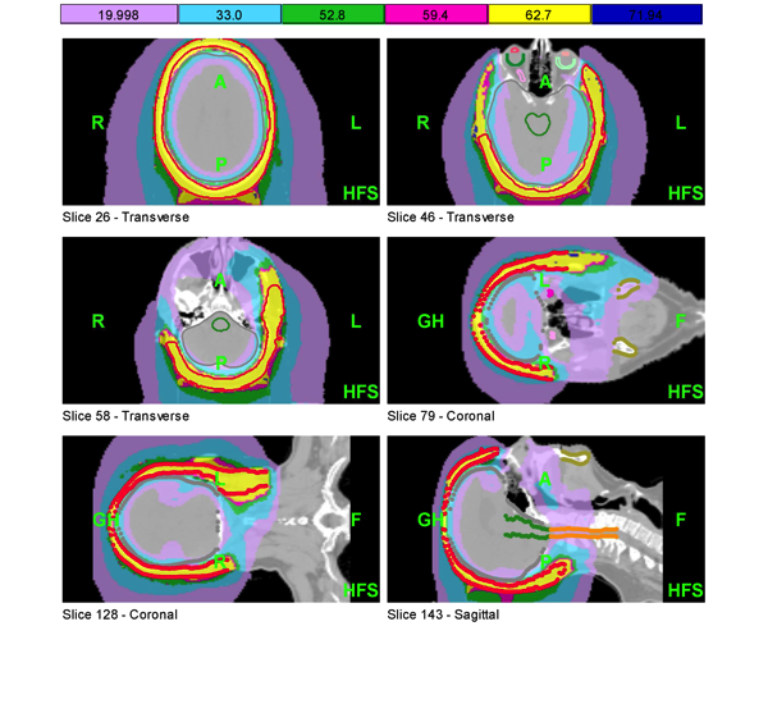
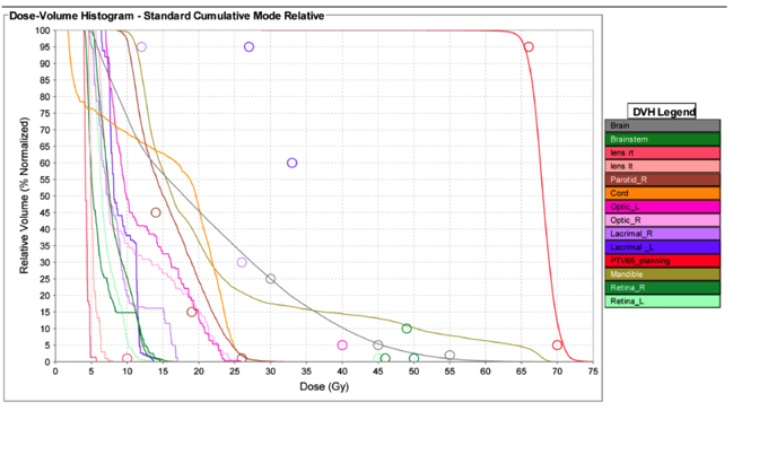
With a high risk of distant and loco-regional recurrence postsurgery, the patient elected to receive adjuvant chemotherapy with weekly paclitaxel (80 mg/m2) for 12 cycles. After completing chemotherapy, the patient received adjuvant radiotherapy (RT). The radiation treatment isodose distribution and the corresponding dose-volume histogram (DVH) are noted in Figures 1 and 2, respectively. The treatment plan was 66 Gy in 33 fractions delivered with helical tomotherapy without bolus and a 5 mm planning target volume (PTV) expansion. This technique is described by Orton, et al.1 Time for delivery of 1 fraction (2 Gy) was 797.2 seconds. Ultimately the patient received 60 Gy in 30 fractions out of concern for viability of his split thickness skin graft. The benefits of using helical tomotherapy on this patient include the ability to treat nodes on the left side of the neck, to avoid field matching issues that can lead to hot and cold spots in the treatment field, and daily megavoltage CT (MVCT) imaging to ensure proper patient setup and accurate dose delivery.
IMAGING FINDINGS AND DIFFERENTIAL DIAGNOSIS
Preoperative magnetic resonance imaging (MRI) of the brain demonstrated a 1.1 × 2.1 × 0.9-cm enhancing lesion in the left anterosuperior parietal scalp with extension to the overlying skin surface. No findings demonstrated intracranial spread. An incidental 6-mm lesion in the right internal-auditory canal likely representing a vestibular schwannoma was noted. Work-up was negative for regional or distant metastatic disease.
Differential diagnosis includes angiosarcoma, basal cell carcinoma, squamous cell carcinoma, Kaposi’s sarcoma, hemangioma, actinic keratosis, and contusion.
DIAGNOSIS
Scalp angiosarcoma
DISCUSSION
As a result of this entity’s rarity, standardized treatment recommendations are primarily based on (A) retrospective institutional series spanning multiple decades that are subject to inherent selection biases and patient/treatment heterogeneity, and (B) extrapolation from treatment algorithms for other soft-tissue sarcoma histologies.2 Complete tumor resection appears to be an important prognostic factor in patients with head-and-neck angiosarcoma. Admittedly, this improved outcome could be due to selection bias of lesions amenable to resection without evidence of spread elsewhere. Additionally, size < 5 cm corresponds with better overall survival and lower locoregional recurrence.3,4 Emerging data suggests that epithelioid morphology is an adverse prognostic factor.3
Scalp angiosarcoma is an aggressive form of angiosarcoma, with high propensity for early metastatic spread and local recurrence. Because of this, field design typically includes total scalp irradiation with large margins and consideration of regional lymphatic treatment.5 The margin size and total coverage area is significantly larger than used to treat other head and neck cancers due to the high rates of local recurrence at the edge of the RT field as well as distant failure. In a Surveillance, Epidemiology and End Results (SEER) database analysis (1973-2007), 5- and 10-year overall survival (OS) rates were noted to be 34% and 14%, respectively.6 Local recurrence rates as noted in different series range from 53% to 100%, while distant metastatic disease in these series ranged from 28% to 64%.7-10 Cutaneous angiosarcomas are the most common subtype of angiosarcoma, and tend to occur in patients ages 65 to 70 with a male predominance.11,12 Angiosarcomas make up 15% of all head-and-neck sarcomas, and approximately 1-2% of all soft-tissue sarcomas.13,14
A wide variety of systemic therapies have been evaluated in angiosarcoma. A phase II study evaluated paclitaxel given as a 60-minute infusion of 80 mg/m2 on days 1, 8, and 15 of a 4-week cycle in metastatic or unresectable angiosarcoma patients. The overall response rate (ORR) at 6 months was 19% with a progression-free survival (PFS) rate at 2 and 4 months of 74% and 45%, respectively. The median overall survival was 8 months.15 Memorial Sloan Kettering Cancer Center (MSKCC), New York, retrospectively evaluated paclitaxel response in 9 scalp angiosarcoma patients, and demonstrated an 89% response rate (4 partial, 4 complete).16 The median duration of response was 5 months (range 2-13 months). These data support the long-held impression that scalp angiosarcomas may be more paclitaxel-responsive than other angiosarcomas (eg, visceral). Activity of pegylated-liposomal doxorubicin in angiosarcoma (all sites) was demonstrated in a series reported from the University of Minnesota, Minneapolis, with 3/6 patients developing a PR lasting 6, 19 and > 20 months.17 Docetaxel resulted in a ORR of 67% (22% complete response [CR]) in a series of 9 cutaneous angiosarcomas.18 A phase II study of bevacizumab in metastatic or locally advanced angiosarcoma and epithelioid hemangioendothelioma demonstrated 4/30 (13%) patients with a partial response (PR), 15/30 (50%) with stable disease, and a mean time to progression of 26 weeks.19 Sorafenib has been shown to have ORR of 14% in angiosarcoma with a median PFS of 3.8 months and median OS of 14.9 months.20 In addition, an Italian retrospective study documented ORR of 68% with gemcitabine in advanced angiosarcoma with a median PFS of 7 months, and OS of 17 months.21
In general, due to a dearth of data, radiation treatment principles for scalp angiosarcoma are extrapolated from the larger soft-tissue sarcoma experience. Routinely used postoperative doses range from 60 to 66 Gy in 1.8 to 2 Gy fractions, with higher doses used when gross disease is present (limited by normal tissue tolerance). A study of 70 patients with nonmetastatic angiosarcoma of the face and scalp found that patients treated with surgery and RT (vs. surgery alone or RT alone) had improved 5-year OS (68% vs. 40% vs. 22%, p = 0.01), disease-specific survival (DSS) (76% vs. 43% vs. 22%, p = 0.006), and better local control (84% vs. 25% vs. 22%, p = 0.0003).10 The benefit of combined modality local therapy remained significant on multivariate analysis (p = 0.0003). This study also found that local recurrence correlated with the development of distant metastatic disease. The University of Florida angiosarcoma experience demonstrated improved outcomes with hyperfractionated (3 times daily) schemes. Nonscalp primary lesions were associated with better outcomes. Improved outcomes were also observed in patients receiving 70 Gy or more, suggesting better outcomes with higher doses of radiation therapy.22 Better outcomes with combined modality therapy have been demonstrated in other studies as well.8,11 Interestingly, Guadagnolo and colleagues found that positive surgical margins did not portend a poorer prognosis, suggesting that aggressive resections that may compromise delivery of timely adjuvant radiation therapy may not be necessary.10 In general, re-excision to obtain negative margins could be beneficial if timely healing is expected, and it is not anticipated to significantly increase morbidity or cosmetic deformity.
For RT delivery, the 3 most commonly used techniques include opposed electron-photon fields, brachytherapy, and intensity-modulated radiation therapy (IMRT). A description of opposed electron-photon fields for scalp irradiation was reported by Akazawa and colleagues,23 and subsequently optimized by investigators at MD Anderson Cancer Center.24 Field matching is done with 6 MV photons and 6 MeV electrons with 2 pairs of lateral electron and photons fields. The electrons treat most of the lateral scalp while photons are used on the superior scalp with a central block to protect underlying brain tissue. Bolus is employed to ensure adequate dose to surface tissues. Due to concerns regarding underdosing at the junction of the photon-electron fields, as originally described, a 3 to 4-mm overlap of the electron and photon fields has been recommended to improve dosimetric homogeneity.
Another technique for scalp irradiation is surface-mold brachytherapy. Ozyar et al. described a technique using remote-controlled afterloading high-dose-rate (HDR) brachytherapy in a patient with multifocal basal cell carcinoma.25 Catheters traversing the entire scalp were placed parallel at 10 to 12-mm spacing. HDR microselection equipment with an Ir-192 source was used. A total dose of 4,050 cGy at 0.5-cm skin depth was given over 3 weeks. The patient remained without evidence of disease through 6 years with no late complications from the radiation. No in vivo dosimetric verification was reported. Advantages of this technique include homogeneous dose distribution with steeper dose fall-off beyond the target, and simpler setup compared to IMRT or opposed photon-electron fields.
A third radiotherapy approach is total scalp irradiation using IMRT.1,26,27 Among the most commonly reported IMRT techniques used is helical tomotherapy. Orton and colleagues have shown that helical tomotherapy has the ability to deliver beamlets tangential to the scalp while avoiding field-matching problems, and only requires the use of one modality. Tangential beamlets available from a tomotherapy unit enhance the dose to superficial tissues, eliminating the need for bolus. Dosimetric verifications of superficial dose were performed on the surface of an anthropomorphic phantom with gafchromic EBT radiochromic film, Kodak EDR2 film, and a skin solid-state dosimeter known as MOSkin. The time for setup, MVCT, shifts and treatment is about 25 minutes, comparable to the time for treatment on a conventional linear accelerator.27 A helical-treatment delivery approach also avoids field-matching problems and allows simultaneous treatment of regional nodal volumes, as was required in our case.
Hadziahmetovic and colleagues recently described a bolus technique called scalp uniform bolus application (SCUBA). Due to concern about daily reproducibility of conventional wax or superflab bolus, they used airtight scalp uniform bolus with 2 wetsuit diving hoods that were equivalent to a 5-mm bolus without air gaps. Optically stimulated luminescent dosimeters (OSLDs) were used to measure dose to the scalp on the head-phantom with either 20-field IMRT or opposed 6 MV photons and 6 MeV electrons. This proves inexpensive, effective, and easily reproducible as an option for uniform bolus for either opposed photon-electron or IMRT plans.28
CONCLUSION
Scalp angiosarcoma is a rare entity with little data to guide treatment decisions. Retrospective studies are difficult to interpret due to heterogeneities in patient selection, disease burden and treatment techniques. For resectable lesions, resection is generally considered the first line of treatment. Given the high propensity for local recurrence, adjuvant radiation therapy is warranted. We have described a variety of radiation approaches to consider, including opposed electron-photon fields, brachytherapy and IMRT, each with advantages and disadvantages. Use of adjuvant chemotherapy also merits consideration in view of the risk for distant recurrences.
REFERENCES
- Orton N, Jaradat H, Welsh J, et al. Total scalp irradiation using helical tomotherapy. Med Dosim. 2005;30:162-168.
- Young RJ, Brown NJ, Reed MW, et al. Angiosarcoma. Lancet Oncol. 2010;11:983-991.
- Lahat G, Dhuka AR, Hallevi H, et al. Angiosarcoma: clinical and molecular insights. Ann Surg. 2010;251:1098-1106.
- Buehler D, Rice SR, Moody JS, et al. Angiosarcoma outcomes and prognostic factors: a 25-year single institution experience. Am J Clin Oncol; 2013. [Epub ahead of print]
- Mendenhall WM, Mendenhall CM, Werning JW, et al. Cutaneous Angiosarcoma. Am J Clin Oncol. 2006;29:524-528.
- Albores-Saavedra J, Schwartz AM, Henson DE, et al. Cutaneous angiosarcoma. Analysis of 434 cases from the Surveillance, Epidemiology, and End Results Program, 1973-2007. Ann Diagn Pathol. 2011;15:93-97.
- Maddox JC, Evans HL. Angiosarcoma of skin and soft tissue: a study of forty-four cases. Cancer. 1981;48:1907-1921.
- Pawlik TM, Paulino AF, McGinn CJ, et al. Cutaneous angiosarcoma of the scalp: A multidisciplinary approach. Cancer. 2003;98:1716-1726.
- Sasaki R, Soejima T, Kishi K, et al. Angiosarcoma treated with radiotherapy: impact of tumor type and size on outcome. Int J Radiat Oncol Biol Phys. 2002;52:1032-1040.
- Guadagnolo BA, Zagars GK, Araujo D, et al. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-667.
- Mark RJ, Tran LM, Sercarz J, et al. Angiosarcoma of the head and neck. The UCLA experience 1955 through 1990. Arch Otolaryngol Head Neck Surg. 1993;119:973-978.
- Lydiatt WM, Shaha AR, Shah JP. Angiosarcoma of the head and neck. Am J Surg. 1994;168:451-454.
- Wanebo HJ, Koness RJ, MacFarlane JK, et al. Head and neck sarcoma: Report of the Head and Neck Sarcoma Registry. Society of Head and Neck Surgeons Committee on Research. Head Neck. 1992; 14:1-7.
- Freedman AM, Reiman HM, Woods JE. Soft-tissue sarcomas of the head and neck. Am J Surg. 1989;158:367-372.
- Penel N, Bui BN, Bay JO, et al. Phase II trial of weekly paclitaxel for unresectable angiosarcoma: the ANGIOTAX Study. J Clin Oncol. 2008;26:5269-5274.
- Fata F, O’Reilly E, Ilson D, et al. Paclitaxel in the treatment of patients with angiosarcoma of the scalp or face. Cancer. 1999;86:2034-207.
- Skubitz KM, Haddad PA. Paclitaxel and pegylated-liposomal doxorubicin are both active in angiosarcoma. Cancer. 2005;104:361-366.
- Nagano T, Yamada Y, Ikeda T, et al. Docetaxel: a therapeutic option in the treatment of cutaneous angiosarcoma: report of 9 patients. Cancer. 2007;110:648-651.
- Agulnik M, Yarber JL, Okuno SH, et al. An open-label, multicenter, phase II study of bevacizumab for the treatment of angiosarcoma and epithelioid hemangioendotheliomas. Ann Oncol. 2013;24:257-263.
- Maki RG, D’Adamo DR, Keohan ML, et al. Phase II study of sorafenib in patients with metastatic or recurrent sarcomas. J ClinOncol. 2009;27:3133-3140.
- Stacchiotti S, Palassini E, Sanfilippo R, et al. Gemcitabine in advanced angiosarcoma: a retrospective case series analysis from the Italian Rare Cancer Network. Ann Oncol. 2012;23:501-508.
- Scott MT, Portnow LH, Morris CG, et al. Radiation therapy for angiosarcoma The 35-year University of Florida Experience. Am J Clin Oncol. 2013;36:174-180.
- Akazawa C. Treatment of the scalp using photon and electron beams. Med Dosim. 1989;14:129-131.
- Tung SS, Shiu AS, Starkschall G, et al. Dosimetric evaluation of total scalp irradiation using a lateral electron-photon technique. Int J Radiat Oncol Biol Phys. 1993;27:153-160.
- Ozyar E, Gurdalli S. Mold brachytherapy can be an optional technique for total scalp irradiation. Int J Radiat Oncol Biol Phys. 2002;54:1286.
- Bedford JL, Childs PJ, Hansen VN, et al. Treatment of extensive scalp lesions with segmental intensity-modulated photon therapy. Int J Radiat Oncol Biol Phys. 2005;62:1549-1558.
- Hardcastle N, Soisson E, Metcalfe P, et al. Dosimetric verification of helical tomotherapy for total scalp irradiation. Med Phys. 2008;35:5061-5068.
- Hadziahmetovic M, Weldon S, Pearson M, et al. Scalp uniform bolus application (SCUBA) technique for homogeneous scalp and regional nodal irradiation. Pract Radiat Oncol. 2014; 4:95-99.