New PET Tracer Enhances Detection of Medullary Thyroid Cancer, Shows Theranostic Potential
 A newly developed PET imaging agent has been found to be effective in identifying medullary thyroid cancer (MTC) in preclinical and clinical studies. The research, published in The Journal of Nuclear Medicine, indicate that the PET imaging agent may be a promising theranostic candidate for clinical use.
A newly developed PET imaging agent has been found to be effective in identifying medullary thyroid cancer (MTC) in preclinical and clinical studies. The research, published in The Journal of Nuclear Medicine, indicate that the PET imaging agent may be a promising theranostic candidate for clinical use.
MTC is one of the rarest forms of thyroid cancer and accounts for approximately three percent of all cases. Since MTC originates from different cells than most thyroid cancers, different imaging and therapy targets are needed for these patients.
“The cholecystokinin-2 receptor (CCK-2R) is overexpressed on most MTC cells and various compounds targeting CCK-2R have been developed over the past several years. Most of these compounds, however, have low metabolic stability, which is not ideal for radioligand therapy,” noted Thomas Günther, PhD, pharmaceutical radiochemist at Stanford University in Stanford, California. “With a simplistic design modification to tackle instability issues, our team created multiple theranostic agents and sought to evaluate their effectiveness.”
In the study, three compounds (DOTA-CCK-66, DOTA-CCK 66.2, and DOTA-MGS5 external reference]) were each labeled separately with 64Cu, 67Ga, and 177Lu. CCK-2R affinity of each of the radiolabeled compounds was examined on MTC cells. All three compounds exhibited a high affinity, however, due to the more favorable in vitro properties overall of DOTA-CCK-66, DOTA-CCK-66.2 was excluded from further studies.
Next, in vivo stability, biodistribution, imaging, and competition studies were performed on mice bearing a CCK-2R-expressing tumor. 68Ga-DOTA-CCK-66 was selected for proof-of-concept PET/CT application based on its overall in vitro and in vivo properties.
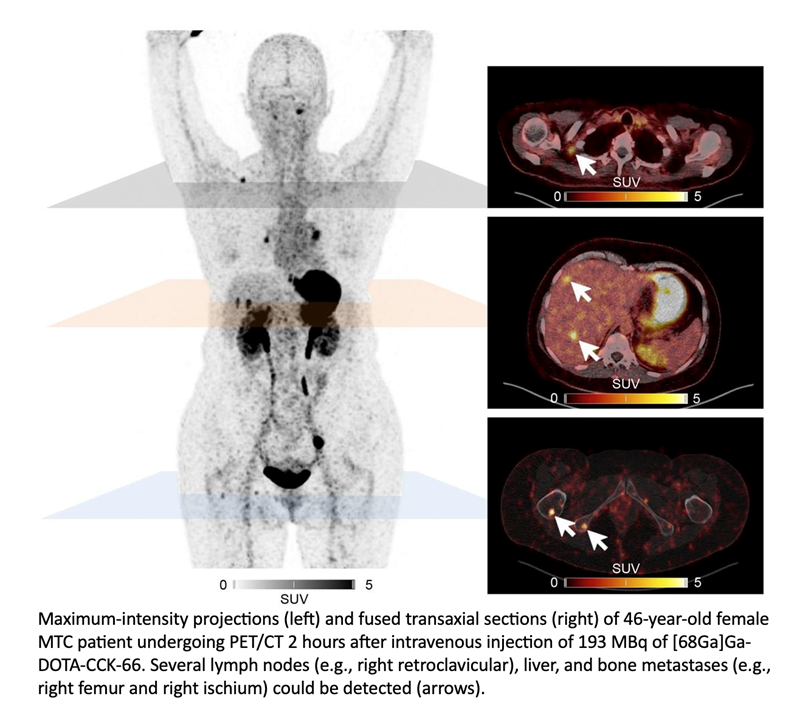
Two MTC patients then underwent 68Ga-DOTA-CCK-66 PET/CT. The compound was well tolerated, showed a favorable biodistribution, and demonstrated high accumulation of activity in tumors.
“Due to increased in vivo stability, our compound reveals favorable tumor uptake as well as an improved activity clearance from off-target tissues. This could result in enhanced lesion detection in PET imaging and additionally enable targeted MTC radioligand therapy,” said Constantin Lapa, MD, director of nuclear medicine at University Hospital Augsburg, in Augsburg, Germany.
Günther and Lapa added, “A significant outcome of our work is the notion that it is possible to optimize pharmacokinetics by chemical design. Analyzing weaknesses of existing compounds and then systematically addressing those to improve imaging and treatment is crucial for future clinical translation.”