Practical Applications of the Internet of Things in Radiation Oncology
Images
SA-CME credits are available for this article here.
Abstract
As the global population ages, there will be an ever-increasing demand on health care systems worldwide for managing chronic conditions, including cancer care. A shift to automated health care solutions will be necessary to improve quality of care while also reducing resource burden to practitioners. Health care systems are amidst a digital transformation from traditional brick-and-mortar care delivery models to those that include virtual care, telehealth, and remote treatment delivery.
The internet of things (IoT) is a system of wireless, interconnected digital devices that can collect, send, and store data over a network without requiring human intervention, and it holds promise of improving the quality of health care while streamlining and enhancing health care delivery. This is especially relevant in technologically oriented medical fields such as radiation oncology. Various applications of IoT have been described in cancer care with immediate relevance to radiation oncology, including the integration of electronic health records (EHR) and nonhealth care data with therapeutic augmented reality, wearable technologies, smart voice assistants, digital medications, artificial intelligence (AI), robots, continuous Bluetooth-enabled monitors, and smart cameras. IoT holds promise of improving primary care through disease prevention and population health initiatives, and improving secondary and tertiary care including cancer care through integration of IoT data to create more coordinated, improved, and proactive care.
However, several challenges to IoT adoption in radiation oncology exist, including the need for more robust policy measures, enhancements in usability and cost effectiveness of IoT devices, improvements in cybersecurity and privacy, transparency of data governance, standardization of protocols to enhance interoperability, and finally, more favorable reimbursement.
Keywords: Internet of Things, IoT, IoMT, therapeutic augmented reality, wearable technologies, smart voice assistants, digital medications, artificial intelligence, robots, smart cameras
The pace of population aging is much faster today than in the past; by 2030, 1 in 6 people worldwide will be aged 60 years or older, and between 2015 and 2050, the proportion of the world’s population over 60 years will nearly double from 12% to 22%.1 As the population ages, there will be an ever-increasing global demand on health care systems for managing chronic conditions, including hypertension, diabetes, obesity, infectious diseases, hematologic disorders, and cancer.2
The cost of cancer care in the US is substantial and rising. The total cost in 2015 was $183 billion and is projected to increase 34% to $246 billion by 2030.3 This expected increase is attributable not only to population growth and increasing life expectancy, but also to suboptimal care coordination, inappropriate or duplicative services, inefficiencies that require outpatient/inpatient follow-up rather than home monitoring, and the high cost of novel therapies.4 This will create major challenges to delivering quality care within our health care system that is safe, timely, effective, efficient, equitable, and patient-centered.5 These demands will ulti- mately require improved population health management techniques and opportunities for enhancing value-based care delivery. The 2020 response to the COVID-19 pandemic has also highlighted the need to transition to technology-based remote health care delivery options.6,7
A shift to automated health care solutions in the information age will be necessary to improve quality of cancer care while also reducing resource burden to practitioners.8 This is particularly important in cancer care where there is a growing emphasis on technologically oriented care delivery such as radiation oncology. IoT includes a world where interconnected internet-enabled devices or “things” can collect and share data (machine-to-machine) without human intervention. In 2020, more than 21 billion devices are estimated to be connected to the internet, and health care IoT (or internet of medical things [IoMT]) could collect health-related data from individuals to improve care delivery and reduce provider burden.9,10 In this review, we provide an overview of the digital transformation of health care systems, IoT technology in cancer care, its practical applications in radiation oncology, and ongoing opportunities and challenges.
The Internet of Things
Digital Transformation of Health Care Systems
Many health care systems are amidst a digital transformation as they move from traditional brick-and-mortar care delivery models to models that include virtual care, telehealth, and remote treatment delivery.11 The COVID-19 pandemic (in 2020 to the present) has especially emphasized the need for a technology-enabled health care system that can facilitate digital transformation.6,7 Health care systems view digital transformation as a way to become more consumer-friendly, but will need to focus on interim milestones to justify value; acquire the talent, data, and key performance indicators needed to overcome digital transformation challenges; and cultivate executive champions.12 However, in a recent survey, only 7 percent of health care and pharmaceutical companies said they had “gone digital,” compared with 15 percent of companies in other industries.13
In response to technological evolutions, IoT technology holds promise to support health care systems to deliver higher quality care and to improve population health initiatives. From a health care perspective, IoT can be considered as any device that can collect health-related data from computing devices, mobile phones, smart bands and wearables, digital medications, implantable surgical devices, or other portable devices that may communicate through channels such as radiofrequency identification that can measure health data and connect to the internet.10 IoMT has been described in health care, with immediate applications to cancer care and radiation oncology, including mobile health, ambient assisted living, wearable devices, smartphones, eHealth, community-based health care and other uses.14 IoT and Healthcare Architecture
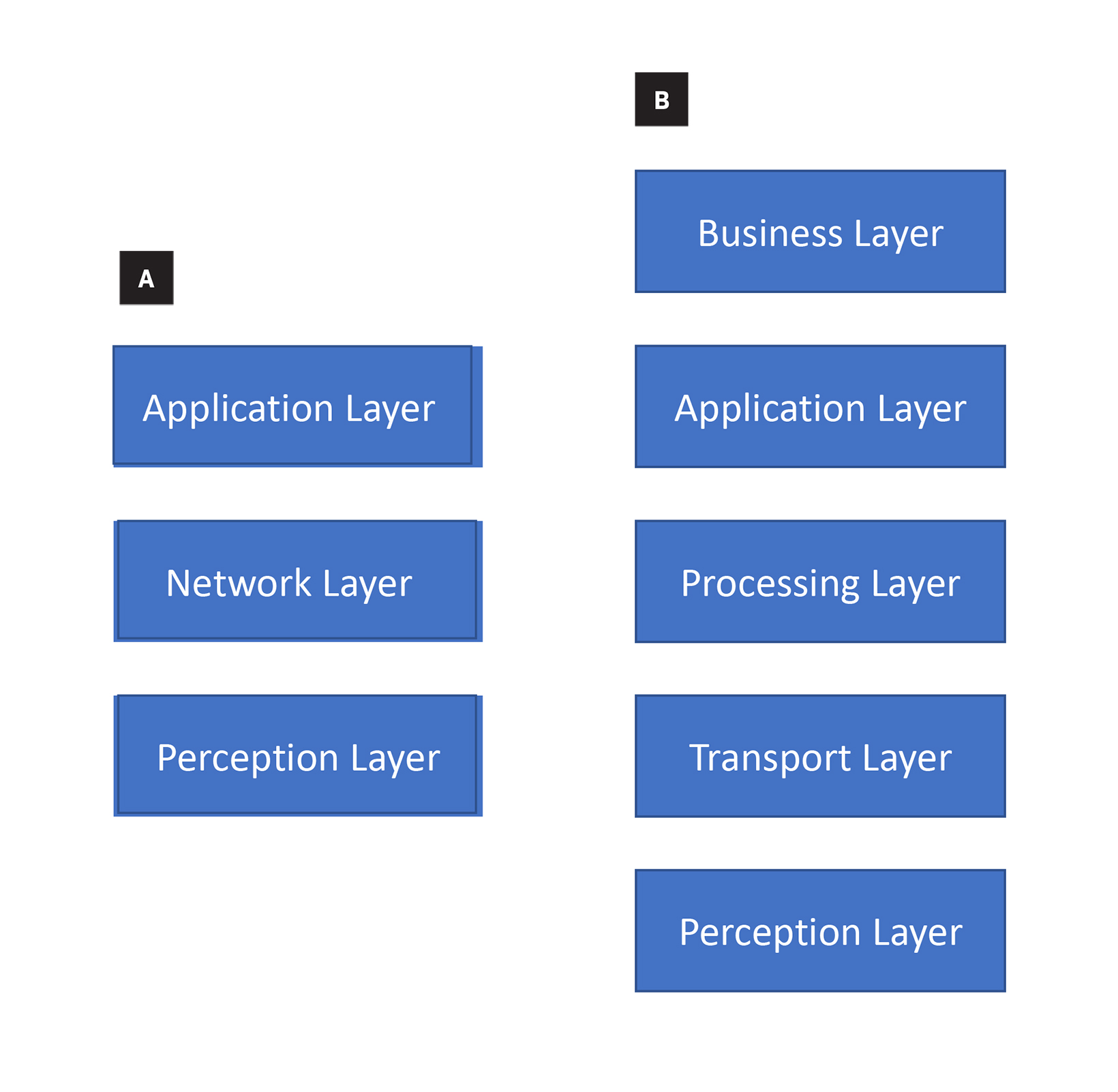
There are 3- and 5-layer IoT architectures that have previously been described and that can be applied to IoT in health care.15-17 Architectures are the way that the components – such as devices, network structure, and cloud technology – are organized. The most basic architecture includes the perception layer, the network layer, and the application layer15,18(Figure 1A), which we will cover briefly here:
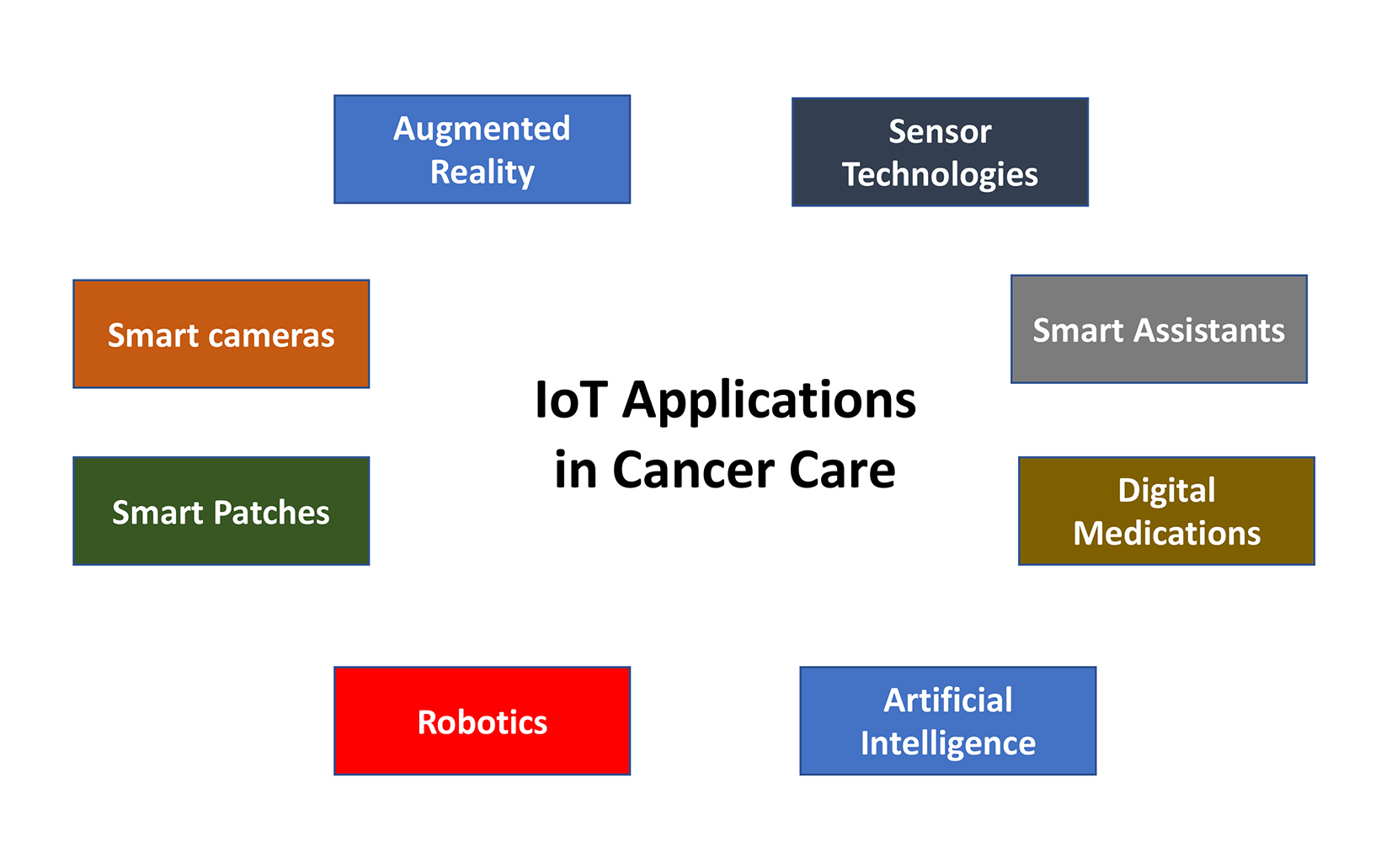
Perception layer. The perception layer is the physical layer that is the foundation of IoT. This layer has sensors for sensing and gathering information about the environment. It senses some physical parameters, identifies other smart objects in the environment, and provides geographic location recognition. This includes radiofrequency identification (RFID), infrared sensors, cameras, GPS, medical sensors, and smart device sensors that can allow for real-time monitoring and network transmission (eg, an implanted, continuous blood glucose level sensor as detailed below). There are numerous examples of IoT devices in health care but limited data on quality and safety.9 Some examples include therapeutic augmented reality, wearable technologies, smart voice assistants, digital medicines, robots, continuous monitors, Bluetooth-enabled monitors, and smart cameras, all of which are discussed in detail below.
Network layer. The network layer is responsible for connecting to other smart things, network devices, and servers. This includes wireless and wired networks that communicate, store, process and transmit sensor data either locally or in a centralized way. Most IoT devices use high frequencies with short-range communication technologies. High-frequency 4G cellular networks have improved potential for communications, and 5G networks are expected to provide a reliable connection for numerous devices simultaneously.19 Centralized cloud-based computing is becoming more popular as it improves flexibility, scalability and access. However, centralization could lead to slower transmission times between central data centers and IoT devices as well as accumulation of unnecessary data. Conversely, the edge cloud allows IoT sensors and network gateways to process and analyze data in a decentralized fashion, reducing the amount of data required to be communicated and managed at a centralized location.20 As an example, blockchain uses decentralized data storage that can be regulated by patients and may improve mobile health applications, monitoring devices, sharing and storing of electronic medical records, clinical trial data, and insurance information storage.21
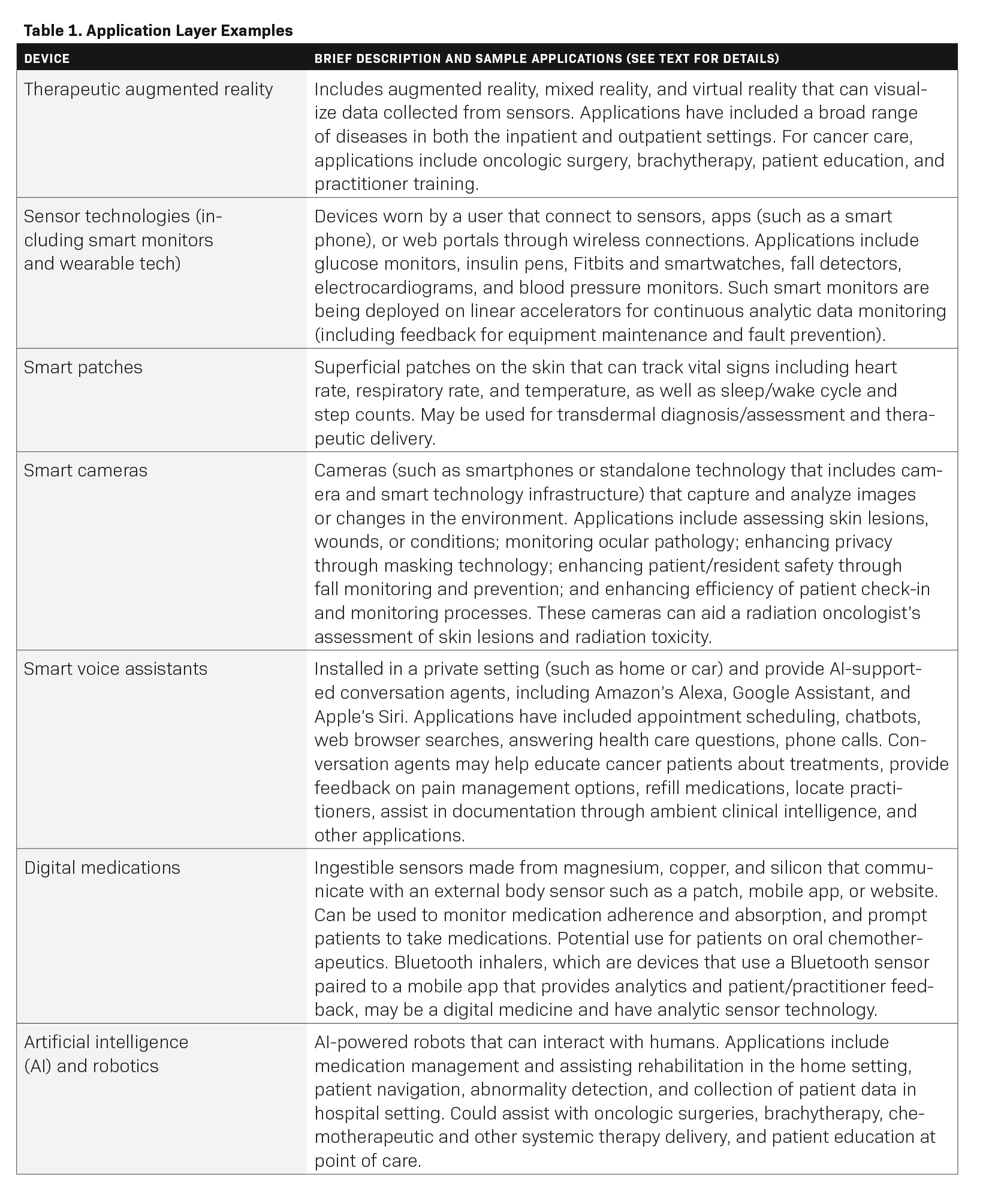
Application layer. The application layer is responsible for delivering application-specific services to the user, such as smart homes, smart cities, and smart health.15 Some examples include therapeutic augmented reality, wearable technologies, smart voice assistants, digital medicines, robots, continuous monitors (eg, an application that records and reads out blood glucose levels from a continuous, implanted sensor), Bluetooth-enabled monitors, and smart cameras (Figure 2), which have immediate applications in radiation oncology. Table 1 lists a brief description of each application, although many others exist.
Research purposes require a more nuanced framework using a 5-layer architecture. The 5-layer architecture adds a transport layer, which transfers the sensor data to and from the perception layer to the processing layer; the processing layer, which stores, analyzes, and processes huge amounts of data that come from the transport layer; and the business layer, which manages applications, business models, and user data/privacy (Figure 1B).
IoT Applications in Cancer Care
Augmented Reality
Therapeutic augmented reality (or extended reality) includes augmented reality (AR), mixed reality (MR), and virtual reality (VR) that can visualize data collected from sensors that are part of the IoT. This technology combines high-quality stereoscopic computer displays such as with goggles to display an immersive 3D environment, with 6 degrees-of-freedom spatial tracking to capture the movements of the user and controllers, and interact with virtual or augmented surroundings.22 Examples have included a broad range of inpatient and outpatient applications to learn about anatomy, anesthesia, central vein catheterization,23 mental health and anxiety disorders,24 stroke,25 pain management,26, 27 and obesity.28 Augmented reality may aid oncologic surgeries,29-31 education for patients undergoing radiation therapy,32,33 immersive virtual reality to reduce patient anxiety and psychological symptoms,26,34 practitioner training,35,36 and brachytherapy.37
Within radiation oncology, AR can provide 3D and 360-degree views to simulate the entire process of radiation therapy, from clinics to simulation rooms and treatment rooms.38 AR will also provide 360-degree views of the treatment room to correct positioning in real-time.39,40 A projector-based display has already been used to simulate controlling a linac for training and education.41< Physicians, dosimetrists, physicists and even patients can explore spatial relationships of dosimetric distribution. For example, a patient with a meningioma may be considering stereotactic radiation therapy and may want to utilize AR to understand the concepts of how the brain and adjacent critical organs at risk may be exposed to radiation due to its proximity to the primary target.
Sensor Technology
Sensor technologies are devices placed on equipment or worn by a user that connect to sensors, apps (such as on a smartphone), or web portals, through wireless connections. Although only 21% of adults and fewer elderly people own wearable devices, the majority of US adults own a smartphone, allowing smartphone technology to rapidly scale IoT-based interventions.42 Applications include continuous glucose monitors, smart insulin pens, loneliness detectors, sleep trackers, smartwatches and Fitbits, fall detectors, wireless electrocardiogram monitors, wearable blood pressure monitors, and others.43-45 Bluetooth inhalers are a related technology that use a Bluetooth sensor paired to a mobile app that provides analytics and patient/practitioner feedback.46
Commonly, wearable devices have been used to assess physical activity levels, as these levels before, during, and after cancer treatment have been established as robust predictors of clinical outcomes as well as quality of life.42,47,48 Interestingly, lower levels of activity during chemoradiation (head and neck, lung, and gastrointestinal cancer) as measured with Garmin devices were associated with greater hospitalization risk, lower likelihood of completing treatment without delays, and shorter survival.49,50 Similarly, daily step count for abdominal cancer patients on postoperative day 7 was inversely correlated with the postoperative complication index.51 Published prospective studies incorporating mobile sensor data with clinical outcomes have focused mostly on patient-reported outcomes, toxicity and symptom burden,51-54quality of life,55 hospitalizations or readmissions,49,50,56,57 or post-operative events.58
Smart monitors are also being deployed on linear accelerators. Such technology allows for continuous background analytic data monitoring that provides feedback for equipment maintenance, proactive service and fault prevention for field service technicians.59 This application has already helped technicians identify early trends in equipment malfunction – such as couch faults or slow multileaf collimator motors – and order and install replacement parts before machine downtime. IoT offers an opportunity to maximize machine uptime and provide personalized, continuous remote support for radiation oncology clinics. Analytics can also be applied to the continuously monitored historical logs and configuration files using machine-learning algorithms.
Smart Patches
Smart patches such as vital sign patches are designed to wirelessly track and monitor heart rate, respiratory rate, sleep cycle, stress levels, temperature, step counts, and falls/incapacitation.60 Temperature-tracking smart patches (TempTraq) are being used in CAR T cell therapy clinical trials.61 In a recent proof-of-concept study, smart patches were used to monitor dyspnea in the palliative care setting.62 Transdermal delivery of chemotherapeutics utilizing smart patches may be a possibility in the future.63 >Smart patches have also been used to biopsy skin cells on the skin surface.64,65 Despite these promising applications, patients may be wary of wearing a patch sensor, and instead opt for biosensors embedded in armbands or wrist-worn devices.66 For example, patients who are receiving concurrent chemoradiation therapy may be at higher risk of hospitalization due to toxicities67 and could benefit from smart patch technology to seamlessly evaluate vital signs and distress in real-time using remote patient monitoring evaluated by a centralized virtual care team.68
Smart Cameras
Smart cameras, such as smartphones or standalone technology that includes camera and smart technology infrastructure, can capture changes in the environment. Smart cameras may support a machine vision system by digitizing and transferring frames for computer analysis although some smart cameras can also serve as self-contained vision systems without relying on external processing equipment.69 Such technology can be used to diagnose, monitor, or evaluate skin conditions including assisting with wound care in patients with diabetes and skin cancer.70-74 Smart cameras can also enhance privacy by using video analytics to hide sensitive health data on displays; enhance patient/resident safety through fall monitoring and prevention; and enhance efficiency of patient check-in, admission, and patient flow through the clinic.75 Within the radiation oncology clinic, smart cameras may capture patient check-in, waiting times, clinic visit times, location of family members, and help monitor safety at the linac and brachytherapy consoles, among other applications.
Smart Voice Assistants
Smart voice assistants can be installed in a private setting (such as the home or car) and provide AI-supported conversation agents, including Amazon’s Alexa, Google Assistant, and Apple’s Siri, to answer a specific set of health-related questions without human contact. Examples include evaluation and management of depression and anxiety; autism; sexual, substance, and physical harassment issues; language impairment; obstructive sleep apnea; hypertension; breast cancer; type 2 diabetes; and pain management.76 Smart voice assistants may also improve access to in-home virtual care.77 Applications have included scheduling appointments; offering chatbots, web browser searches, and phone calls; educating cancer patients about treatments; providing feedback on pain management options; refilling medications; locating practitioners; assisting with documentation through ambient clinical intelligence; and other applications.78-82 Specifically for radiation oncology, a smart voice assistant could remind patients about the day of their weekly treatment management visit, the time of their scheduled daily radiation treatment (with special reminders when schedules change); record notes prior to, during, and after visits; and help find high-quality education for patients and caregivers.
Digital Medications
Digital medications include an ingestible sensor (typically made from magnesium, copper, or silicon) that can communicate with an external body sensor such as a wearable patch or mobile app. Information is stored on the cloud and used to measure medication adherence, absorption, activity, and heart rate.83 In 2019, Proteus Digital Health introduced digital chemotherapy for stage 3 or stage 4 colorectal cancer.84 When a patient swallows the capsule, a sensor activates when it reaches the stomach, which then transmits data to a smart patch with the time of day, the dose, and the type of medication. Unfortunately, Proteus recently filed for bankruptcy, driven by an expensive technology without a clear business model and uneven patient acceptance of ingestible sensors.85 This example illustrates the importance of demonstrating a clear value proposition and product-market fit. In radiation oncology, these technologies (linked with smart patches) can assess patient compliance and toxicities with concurrent chemotherapies (such as capecitabine or temozolomide).
Artificial Intelligence
Overall, AI is a promising medical application leveraging IoT. Narrow AI applications include natural language processing, image analysis, drug discovery development, and computational genomics. Recently, Google Health and Meditech have announced a collaboration to improve clinical search functionality in Expanse electronic health record, which would be another practical use of AI technology.86 With data generated from mobile health apps and IoT devices, AI and deep learning can also optimize disease management and provide big data analysis.86 Big data in health care has rapidly grown to include genomics, metabolomics, proteomics, lipomics, transcriptomics, immunomics, glycomics and imageomics.88-90
Deep learning, a subset of AI, is now being used in decision-making, autosegmentation, radiation treatment planning, and adaptive radiation therapy, but may be limited by access to the internet, web-based cloud solutions, or high-performance computing hardware; and lack of protocols for clinical commissioning, validation, implementation, and maintenance.91 Ultimately, AI holds promise of augmenting or improving efficiencies rather than replacing radiation oncologists’ toolsets, although it may refocus tasks performed by the treatment planning team.91,92
Robotics
Robots using AI technology can interact with humans in various health care settings. Examples have included robots assisting patients with hospital navigation, collecting patient data and assisting with physical therapy and rehabilitation.93,94 Robotic surgery has gained acceptance in minimally invasive surgery,95 and AI in surgery has shaped preoperative planning and intraoperative guidance.96,97 AI-powered robotics could also have applications in brachytherapy, including placement of applicators/needles, and chemotherapeutic and other systemic therapy delivery.
IoT and Improving Cancer Care Delivery
IoT can enable smart devices to transform traditional cancer care in the radiation oncology clinic into a more efficient, higher-quality, technology-enabled service. IoT will be able to impact health care delivery across the full spectrum of care delivery, from primary care to tertiary/quaternary care.
Oncology patients are not only grappling with acute care for their cancer diagnosis but also with competing risks with comorbidities. Primary care focuses on disease prevention, reducing disease burden, modifying risk factors, and caring for patient populations. Unfortunately, 28% of men and 17% of women do not have a primary care provider and lack chronic care services, which will also impact compliance and outcomes from oncologic therapies.98 IoT will enable patients to better self-manage conditions and allow providers to assess modifiable risk factors in real-time.99 It will also increase capacity and access to primary care using AI (including chatbots) and smart voice assistants. However, there is no well-defined pathway for regulatory approval of smart apps, and data on safety and efficacy is lacking.100,101 IoT will also be able to help integrate nonhealth data through data from smart homes, including data from independent living units, grocery/refrigeration, wearable devices, power, appliances, security, and entertainment, to provide a more holistic view of patients and their unique needs. When applied over the population level, it will enhance disease surveillance and assessment of environmental risk factors. During the COVID-19 pandemic, big data analytics have been applied to credit card payments, television surveillance, and geographic location to contact trace and identify close contacts.102
IoT will also directly improve cancer care delivery. Better integration of IoT data with EHRs and nonhealth data will lead to more coordinated and proactive care, as opposed to the current piecemeal, uncoordinated care within the traditional health care system. For instance, IoT can better connect physicians with patients in their home, allowing physicians to better understand patients, better assess toxicities from systemic therapies and radiation, assess treatment compliance, reduce costs, and improve quality of care. IoT will also encourage self-monitoring of data uploaded to the cloud and allow for continuous feedback from providers; create alerts when intervention is needed; help emergency departments and urgent care facilities better triage patients; and increase the number of point-of-care tests rather than outsource to centralized locations.8,10,103
Within the radiation oncology clinic, IoT will allow for broader communication with cloud platforms, data centers, and remote monitoring and control systems. It will facilitate clinic workflows, automation of narrow tasks such as contouring,104,105 treatment and adaptive planning,106,107 quality assurance and quality control procedures,108,109 patient positioning with sensors, intelligent image-guided radiation therapy and intelligent robotics, and communication with radiation information systems potentially even in remote settings with limited availability of trained on-site staffing. Data from smart homes can be integrated with wearable sensors and digital medications/pillboxes to provide contextual data to radiation oncologists and multidisciplinary teams.
Challenges
To further support the growth and use of IoT in cancer care delivery, health care policy will need to support mobile and eHealth technologies. This will require government and private sector investment in IoT hardware and software infrastructure and a transparent and efficient regulatory pathway for approval with the US Food and Drug Administration (FDA). Several countries already have policies for IoT.110 Despite ongoing evidence development in other industries, there are still limited use cases in health care, and limited government (including FDA) regulations and approval.
IoT technology will also need to become more usable and cost effective for consumers of all socioeconomic backgrounds in order to increase user acceptability and confidence in the technology. For instance, a much lower percentage of 55- to 64-year-olds use smart devices compared with those ages 18 to 29 years.111 Only 15% of Americans with an annual household income below $30,000 own a smart speaker while that number rises to 34% for those who earn above $75,000.111 There is also limited understanding from both patients and physicians of the value of cloud-based storage systems, an unfamiliarity with IoT technology, and limited organizational readiness to adopting such technology.112 Patients and physicians may also be reluctant to adopt these technologies if they are intrusive, difficult to use with current daily schedules or workflows, or associated with an overwhelming amount of data. Furthermore, cybersecurity risks remain an obstacle of growth and integration of IoT technologies with existing technologies, especially as the number of entry points with each internet-connected device increases. This is especially the case in health care where protected health information (PHI) is being transmitted across machines and must abide by the Health Insurance Portability and Accountability Act of 1996 (HIPAA). IoT communications are wireless and most utilize low energies, both of which increase the difficulty in ensuring security. Although draft security feature recommendations for IoT devices have been released, the level to which these will be adopted or how they can be enforced is unclear.113
Transparency of data governance and ownership will also be needed for IoT applications. Cloud-based aggregation of IoT data has resulted in centralized cloud storage, which has raised questions about who owns the health care data, and who can view, edit or delete the data. Sharing of this data between states, nations, and organizations is also an important consideration.
Lack of standardization of protocols has also created issues about interoperability of IoT devices with each other or existing legacy technologies. There is not yet consensus regarding wireless communication protocols and standards for machine-to-machine communication. Even existing EHR technology lacks full semantic interoperability, although a push toward Fast Healthcare Interoperability Resources (FHIR) holds promise.114
Finally, there may be an increased resource and information burden to health care providers without adequate reimbursement. In traditional health care systems, health care staff lack expertise to assist with data monitoring and there is an increased burden to clinicians for reviewing large volumes of data. There may also be increased malpractice liability associated with data monitoring. The COVID-19 pandemic has improved reimbursement for telehealth services (commensurate with its increased utilization), but reimbursement for IoT applications will need a concerted effort across governmental and private payers.115
Summary and Conclusions
IoT applications hold great potential to improve the quality and efficiency of cancer care with several practical applications to radiation oncology. As health care systems transform from traditional care delivery models to digital health models, IoT will enable integration of EHR and nonhealth care data with therapeutic augmented reality, wearable technologies, smart voice assistants, digital medicines, robots with AI capabilities, continuous and Bluetooth-enabled monitors, and smart cameras. However, implementation and full realization of the value of IoT will require more robust policy measures, enhancements in usability and cost effectiveness of IoT devices, improvements in cybersecurity and privacy, transparency of data governance, standardization of protocols to enhance interoperability, and more favorable reimbursement. These innovations will improve disease prevention and population health initiatives as well as high-acuity care such as cancer care.
References
- World Health Organization. Ageing and health. 2021. Accessed July 4, 2022. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789-1858.
- Mariotto AB, Enewold L, Zhao J, Zeruto CA, Yabroff KR. Medical care costs associated with cancer survivorship in the United States. Cancer Epidemiol Biomarkers Prev. 2020;29:1304-1312.
- Osarogiagbon RU, Mullangi S, Schrag D. Medicare spending, utilization, and quality in the oncology care model. JAMA. 2021;326:1805-1806.
- Richards MK, Goldin AB. Patient-centered care and quality: activating the system and the patient. Semin Pediatr Surg. 2015;24:319-322.
- Monaghesh E, Hajizadeh A. The role of telehealth during COVID-19 outbreak: a systematic review based on current evidence. BMC Public Health. 2020;20:1193.
- Lee P, Abernethy A, Shaywitz D, et al. Digital health COVID-19 impact assessment: lessons learned and compelling needs. NAM Perspect. 2022;2022.
- Mitchell M, Kan L. Digital technology and the future of health systems. Health Syst Reform. 2019;5:113-20.
- Kelly JT, Campbell KL, Gong E, Scuffham P. The internet of things: impact and implications for health care delivery. J Med Internet Res. 2020;22:e20135.
- Dang LM, Piran MJ, Han D, Min K, Moon H. A survey on internet of things and cloud computing for healthcare. Electronics. 2019:768.
- Torous J, Jan Myrick K, Rauseo-Ricupero N, Firth J. Digital mental health and COVID-19: using technology today to accelerate the curve on access and quality tomorrow. JMIR Ment Health. 2020;7:e18848.
- Appleby C, Wurz J, Hendricks J, Shudes C. Digital transformation. 2021. Accessed July 4, 2022. https://www2.deloitte.com/us/en/insights/industry/health-care/ digital-transformation-in-healthcare.html
- Reddy M. Digital Transformation in Healthcare in 2022: 7 Key Trends. 2022. Accessed July 4, 2022. https://www.digitalauthority.me/resources/state-of-digital-transformation-healthcare/
- Shah Nazir YA, Naeem Ullah, Iván García-Magariño. Internet of things for healthcare using effects of mobile computing: a systematic literature review. Wireless Com Mobile Comp. 2019;2019:20.
- Sethi P, Sarangi SR. Internet of things: architectures, protocols, and applications. J Elec Comp Eng. 2017;2017:9324035.
- Mashal I, Alsaryrah O, Chung T-Y, Yang C-Z, Kuo W-H, Agrawal DP. Choices for interaction with things on Internet and underlying issues. Ad Hoc Net. 2015;28:68-90.
- Miao W, Ting-Jie L, Fei-Yang L, Jing S, Hui-Ying D. Research on the architecture of internet of things. 2010 3rd International Conference on Advanced Computer Theory and Engineering (ICACTE). 2010;V5-484-V5-7.
- Said O, Masud M. Towards internet of things: survey and future vision. Int J Comp Net. 2013;5:1-17.
- Li S, Xu LD, Zhao S. 5G Internet of things: a survey. J Indust Info Integrat. 2018;10:1-9.
- Pan J, McElhannon J. Future edge cloud and edge computing for internet of things applications. IEEE Intern Things J. 2018;5:439-449.
- Chen HS, Jarrell JT, Carpenter KA, Cohen DS, Huang X. Blockchain in healthcare: a patient-centered model. Biomed J Sci Tech Res. 2019;20:15017-22.
- Williams CL, Kovtun KA. The future of virtual reality in radiation oncology. Int J Radiat Oncol Biol Phys. 2018;102:1162-1164.
- Gerup J, Soerensen CB, Dieckmann P. Augmented reality and mixed reality for healthcare education beyond surgery: an integrative review. Int J Med Ed. 2020;11:1-18.
- Valmaggia LR, Latif L, Kempton MJ, Rus-Calafell M. Virtual reality in the psychological treatment for mental health problems: a systematic review of recent evidence. Psychiat Res. 2016;236:189-195.
- Lohse KR, Hilderman CG, Cheung KL, Tatla S, Van der Loos HF. Virtual reality therapy for adults post-stroke: a systematic review and meta-analysis exploring virtual environments and commercial games in therapy. PLoS One. 2014;9:e93318.
- Chirico A, Lucidi F, De Laurentiis M, Milanese C, Napoli A, Giordano A. Virtual reality in health system: beyond entertainment. a mini-review on the efficacy of VR during cancer treatment. J Cell Physiol. 2016;231:275-287.
- Tashjian VC, Mosadeghi S, Howard AR, et al. Virtual reality for management of pain in hospitalized patients: results of a controlled trial. JMIR Ment Health. 2017;4:e9.
- Persky S. Application of virtual reality methods to obesity prevention and management research. J Diabetes Sci Technol. 2011;5:333-339.
- Gouveia PF, Costa J, Morgado P, et al. Breast cancer surgery with augmented reality. Breast. 2021;56:14-17.
- Sahovaler A, Chan HHL, Gualtieri T, et al. Augmented reality and intraoperative navigation in sinonasal malignancies: a preclinical study. Front Oncol. 2021;11:723509.
- Tagaytayan R, Kelemen A, Sik-Lanyi C. Augmented reality in neurosurgery. Arch Med Sci. 2018;14:572-578.
- Wang LJ, Casto B, Luh JY, Wang SJ. Virtual reality-based education for patients undergoing radiation therapy. J Cancer Ed. 2022;37:694-700.
- van der Kruk SR, Zielinski R, MacDougall H, Hughes-Barton D, Gunn KM. Virtual reality as a patient education tool in healthcare: a scoping review. Patient Educ Couns.2022;105:1928-1942.
- Reynolds LM, Cavadino A, Chin S, et al. The benefits and acceptability of virtual reality interventions for women with metastatic breast cancer in their homes; a pilot randomised trial. BMC Cancer. 2022;22:360.
- Herbst R, Rybak T, Meisman A, et al. A virtual reality resident training curriculum on behavioral health anticipatory guidance: development and usability study. JMIR Ped Parent. 2021;4:e29518.
- Hooper J, Tsiridis E, Feng JE, et al. Virtual reality simulation facilitates resident training in total hip arthroplasty: a randomized controlled trial. J Arthroplasty. 2019;34:2278-2283.
- Taunk NK, Shah NK, Hubley E, Anamalayil S, Trotter JW, Li T. Virtual reality-based simulation improves gynecologic brachytherapy proficiency, engagement, and trainee self-confidence. Brachytherapy. 2021;20:695-700.
- Penn Radiation Oncology VR Experience. 2022. Accessed July 4, 2022. https://www.oncolink.org/penn-radiation-oncology-vr-experience
- Johnson K, Liszewski B, Dawdy K, Lai Y, McGuffin M. Learning in 360 degrees: a pilot study on the use of virtual reality for radiation therapy patient educa- tion. J Med Imag Radiat Sci. 2020;51:221-226.
- Chamunyonga C, Burbery J, Caldwell P, Rutledge P, Fielding A, Crowe S. Utilising the virtual environment for radiotherapy training system to support under- graduate teaching of IMRT, VMAT, DCAT treatment planning, and QA concepts. J Med Imag Radiat Sci. 2018;49:31-38.
- Kane P. Simulation-based education: a narrative review of the use of VERT in radiation therapy education. J Med Imag Radiat Sci. 2018;65:131-136.
- Low CA. Harnessing consumer smartphone and wearable sensors for clinical cancer research. NPJ Digit Med. 2020;3:140.
- Sangave NA, Aungst TD, Patel DK. Smart connected insulin pens, caps, and attachments: a review of the future of diabetes technology. Diabetes Spectr. 2019;32:378-384.
- Doryab A, Villalba DK, Chikersal P, et al. Identifying behavioral phenotypes of loneliness and social isolation with passive sensing: statistical analysis, data mining and machine learning of smartphone and Fitbit data. JMIR Mhealth Uhealth. 2019;7:e13209.
- Kakria P, Tripathi NK, Kitipawang P. A real-time health monitoring system for remote cardiac patients using smartphone and wearable sensors. Int J Telemed Appl. 2015;2015:373474.
- Merchant RK, Inamdar R, Quade RC. Effectiveness of population health management using the propeller health asthma platform: a randomized clinical trial. J Allergy Clin Immunol Pract. 2016;4:455-463.
- Ballard-Barbash R, Friedenreich CM, et al. Physical activity, biomarkers, and disease outcomes in cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104:815-840.
- Ferrer RA, Huedo-Medina TB, Johnson BT, Ryan S, Pescatello LS. Exercise interventions for cancer survivors: a meta-analysis of quality of life outcomes. Ann Behav Med. 2011;41:32-47.
- Ohri N, Halmos B, Bodner WR, et al. Daily step counts: a new prognostic factor in locally advanced non-small cell lung cancer? Int J Radiat Oncol Biol Phys. 2019;105:745-751
- Ohri N, Kabarriti R, Bodner WR, et al. Continuous activity monitoring during concurrent chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2017;97:1061-1065.
- Sun V, Dumitra S, Ruel N, et al. Wireless monitoring program of patient-centered outcomes and recovery before and after major abdominal cancer surgery. JAMA Surg. 2017;152:852-859
- Bennett AV, Reeve BB, Basch EM, et al. Evaluation of pedometry as a patient-centered outcome in patients undergoing hematopoietic cell transplant (HCT): a comparison of pedometry and patient reports of symptoms, health, and quality of life. Qual Life Res. 2016;25:535-546.
- Soto-Perez-De-Celis E, Kim H, Rojo-Castillo MP, et al. A pilot study of an accelerometer-equipped smartphone to monitor older adults with cancer receiving chemotherapy in Mexico. J Geriat Oncol. 2018;9:145-151.
- Wright AA, Raman N, Staples P, et al. The HOPE Pilot Study: harnessing patient-reported outcomes and biometric data to enhance cancer care. JCO Clin Cancer Inform. 2018;2:1-12.
- Bade BC, Brooks MC, et al. Assessing the correlation between physical activity and quality of life in advanced lung cancer. Integrat Cancer Ther. 2018;17:73-79.
- Low CA, Bovbjerg DH, Ahrendt S, et al. Fitbit step counts during inpatient recovery from cancer surgery as a predictor of readmission. Ann Behav Med. 2018;52:88-92.
- Pavic M, Klaas V, Theile G, et al. Mobile health technologies for continuous monitoring of cancer patients in palliative care aiming to predict health status deterioration: a feasibility study. J Palliat Med. 2020;23:678-685.
- Panda N, Solsky I, Huang EJ, et al. Using smartphones to capture novel recovery metrics after cancer surgery. JAMA Surg. 2020;155:123-129.
- Less Downtime, More Insights with Varian Smart Services. 2022. Accessed July 4, 2022. https://www.varian.com/resources-support/blogs/clinical-oncology- news/less-downtime-more-insights-varian-smart-services
- Levine DM, Ouchi K, Blanchfield B, et al. Hospital-level care at home for acutely ill adults: a pilot randomized controlled trial. J Gen Intern Med. 2018;33:729-736.
- Brown B. Temperature Tracking Smart Patch Aids New Cancer Treatment Trials. 2021. Accessed July 4, 2022. https://healthtechinsider.com/2021/06/30/tem- perature-tracking-smart-patch-aids-new-cancer-treatment-trials/
- Mallick MK, Biser S, Haridas A, et al. Improving dyspnoea symptom control of patients in palliative care using a smart patch – a proof of concept study. Front Dig Health. 2021;3:765867.
- Neupane R, Boddu SHS, Abou-Dahech MS, et al. Transdermal delivery of chemotherapeutics: strategies, requirements, and opportunities. Pharmaceutics. 2021;13.
- Gerami P, Yao Z, Polsky D, et al. Development and validation of a noninvasive 2-gene molecular assay for cutaneous melanoma. J Am Acad Dermatol. 2017;76:114-20.e2.
- DermTech. 2022. Accessed July 4, 2022. https://dermtech.com/
- Wong CK, Ho DTY, Tam AR, et al. Artificial intelligence mobile health platform for early detection of COVID-19 in quarantine subjects using a wearable bio-sensor: protocol for a randomised controlled trial. BMJ Open. 2020;10:e038555.
- Tonse R, Ramamoorthy V, Rubens M, et al. Hospitalization rates from radiotherapy complications in the United States. Sci Rep. 2022;12:4371.
- Pritchett JC, Borah BJ, Desai AP, et al. Association of a remote patient monitoring (RPM) program with reduced hospitalizations in cancer patients with COVID-19. JCO Oncol Pract. 2021;17:e1293-e302.
- Smart Camera Integration in Machine Vision Systems. 2022. Accessed July 4, 2022. https://www.thomasnet.com/articles/automation-electronics/smart-camera-machine-vision-system/
- Ashique KT, Kaliyadan F, Aurangabadkar SJ. Clinical photography in dermatology using smartphones: an overview. Indian Dermatol Online J. 2015;6:158-163.
- Bhelonde A, Didolkar N, Jangale S, Kulkarni NL. Flexible wound assessment system for diabetic patient using android smartphone. 2015 International Conference on Green Computing and Internet of Things (ICGCIoT); 2015;466-469.
- Rat C, Hild S, Rault Sérandour J, et al. Use of smartphones for early detection of melanoma: systematic review. J Med Internet Res. 2018;20:e135.
- Das K, Cockerell CJ, Patil A, et al. Machine learning and its application in skin cancer. Int J Environ Res Public Health. 2021;18.
- Wu Y, Chen B, Zeng A, Pan D, Wang R, Zhao S. Skin cancer classification with deep learning: a systematic review. Front Oncol. 2022;12:893972.
- Thorpe J. Three ways healthcare can be improved with smart cameras. 2021. Accessed July 4, 2022. https://internationalsecurityjournal.com/health-care-smart-cameras/
- Laranjo L, Dunn AG, Tong HL, et al. Conversational agents in healthcare: a systematic review. J Am Med Inform Assoc. 2018;25:1248-58.
- Teladoc Health and Amazon Team Up to Launch Teladoc on Alexa. 2022. Accessed July 4, 2022. https://www.teladochealth.com/newsroom/press/release/ Teladoc-Health-and-Amazon-Team-Up-to-Launch-Teladoc-on-Alexa/
- McDonald R. ‘Alexa For Health Care’: New Virtual Assistant Helps Patients with Cancer at Home. 2020. Accessed July 4, 2022. https://www.curetoday.com/ view/alexa-for-health-care-new-virtual-assistant-helps-patients-with-cancer-at-home
- Schwartz E. New Portable Smart Speaker Helps Oncologists Treat Cancer Patients. 2021. Accessed July 4, 2022. https://voicebot.ai/2021/01/19/new-portable- smart-speaker-helps-oncologists-treat-cancer-patients/
- Baertsch M-A, Decker S, Probst L, et al. Convenient access to expert-reviewed health information via an Alexa voice assistant skill for patients with multiple myeloma: development study. JMIR Cancer. 2022;8:e35500.
- Martinez-Martin N, Luo Z, Kaushal A, et al. Ethical issues in using ambient intelligence in health-care settings. Lancet. Dig Health. 2021;3:e115-e23.
- Avendano JP, Gallagher DO, Hawes JD, et al. Interfacing with the electronic health record (EHR): a comparative review of modes of documentation. Cureus. 2022;14:e26330.
- Plowman RS, Peters-Strickland T, Savage GM. Digital medicines: clinical review on the safety of tablets with sensors. Expert Opin Drug Saf. 2018;17:849-852.
- Robbins R. A ‘digital pill’ for cancer patients is rolled out for the first time, in hopes of improving outcomes. 2019. Accessed July 4, 2022. https://www.statnews. com/2019/01/17/a-digital-pill-for-cancer-patients-is-rolled-out-for-the-first-time-in-hopes-of-improving-outcomes/
- Landi H. From big deals to bankruptcy, a digital health unicorn falls short. Here’s what other startups can learn from Proteus. 2020. Accessed July 4, 2022. https://www.fiercehealthcare.com/tech/from-billions-to-bankruptcy-proteus-digital-health-fell-short-its-promise-here-s-what-other
- Siwicki B. Meditech and Google Health to collaborate on clinical search in Expanse EHR. 2022. Accessed July 4, 2022. https://www.healthcareitnews.com/ news/meditech-and-google-health-collaborate-clinical-search-expanse-ehr
- Tobore I, Li J, Yuhang L, et al. Deep learning intervention for health care challenges: some biomedical domain considerations. JMIR Mhealth Uhealth. 2019;7:e11966.
- Morrison JJ, Hostetter J, Wang K, Siegel EL. Data-driven decision support for radiologists: re-using the national lung screening trial dataset for pulmonary nodule management. J Dig Imag. 2015;28:18-23.
- Chen RC, Gabriel PE, Kavanagh BD, McNutt TR. How will big data impact clinical decision making and precision medicine in radiation therapy? Int J Radiat Oncol Biol Phys. 2016;95:880-884.
- Chen G, Cui J, Qian J, et al. Rapid progress in intelligent radiotherapy and future implementation. Cancer Investig. 2022;40:425-436.
- Netherton TJ, Cardenas CE, Rhee DJ, Court LE, Beadle BM. The emergence of artificial intelligence within radiation oncology treatment planning. Oncology. 2021;99:124-134.
- Mesko B. Doctors Reject Support From A.I. – Here’s Why. 2021. Accessed July 4, 2022. https://www.linkedin.com/pulse/doctors-reject-support-from-ai-heres- why-bertalan-mesk%C3%B3-md-phd/
- Wan S, Gu Z, Ni Q. Cognitive computing and wireless communications on the edge for healthcare service robots. Comp Com. 2020;149:99-106.
- Putte Dvd, Boumans R, Neerincx M, Rikkert MO, Mul Md. A social robot for autonomous health data acquisition among hospitalized patients: an exploratory field study. 2019 14th ACM/IEEE International Conference on Human-Robot Interaction (HRI); 2019;658-659.
- Vijayakumar M, Shetty R. Robotic surgery in oncology. Indian J Surg Oncol. 2020;11:549-551.
- Kwo L. Contributed: The power of AI in surgery. 2021. Accessed July 4, 2022. https://www.mobihealthnews.com/news/contributed-power-ai-surgery
- Feizi N, Tavakoli M, Patel RV, Atashzar SF. Robotics and AI for teleoperation, tele-assessment, and tele-training for surgery in the era of COVID-19: existing challenges, and future vision. Front Robot AI. 2021;8:610677.
- Finnegan J. Many Americans don’t have a primary care doctor. 2017. Accessed July 4, 2022. https://www.fiercehealthcare.com/practices/many-americans-don-t-have-a-primary-care-doctor
- Vickery DM, Kalmer H, Lowry D, Constantine M, Wright E, Loren W. Effect of a self-care education program on medical visits. JAMA. 1983;250:2952-2956.
- Borycki E. Quality and safety in eHealth: the need to build the evidence base. J Med Internet Res. 2019;21:e16689.
- Bates DW, Landman A, Levine DM. Health apps and health policy: What is needed? JAMA. 2018;320:1975-1976.
- Chen CM, Jyan HW, Chien SC, et al. Containing COVID-19 among 627,386 persons in contact with the diamond princess cruise ship passengers who disembarked in Taiwan: big data analytics. J Med Internet Res. 2020;22:e19540.
- Palmieri F, Ficco M, Pardi S, Castiglione A. A cloud-based architecture for emergency management and first responders localization in smart city environments. Comput Elec Engin. 2016;56:810-830.
- Siddique S, Chow JCL. Artificial intelligence in radiotherapy. Reports Pract Oncol Radiother. 2020;25:656-666.
- Song Y, Hu J, Wu Q, et al. Automatic delineation of the clinical target volume and organs at risk by deep learning for rectal cancer postoperative radiotherapy. Radiother Oncol. 2020;145:186-192.
- Fung NTC, Hung WM, Sze CK, Lee MCH, Ng WT. Automatic segmentation for adaptive planning in nasopharyngeal carcinoma IMRT: time, geometrical, and dosimetric analysis. Med Dosim. 2020;45:60-65.
- Han EY, Kim GY, Rebueno N, Yeboa DN, Briere TM. End-to-end testing of automatic plan optimization using RayStation scripting for hypofractionated multi-metastatic brain stereotactic radiosurgery. Med Dosim. 2019;44:e44-e50.
- McNutt TR, Moore KL, Wu B, Wright JL. Use of big data for quality assurance in radiation therapy. Sem Rad Onc. 2019;29:326-332.
- Kalet AM, Luk SMH, Phillips MH. Radiation therapy quality assurance tasks and tools: the many roles of machine learning. Med Phys. 2020;47:e168-e77.
- Global Internet of Things Policies 2018-2019. 2019. Accessed July 4, 2022. https://www.globenewswire.com/news-release/2019/05/30/1859482/0/en/Global- Internet-of-Things-Policies-2018-2019.html
- Stasha S. Amazon Alexa Statistics, Facts, and Trends. 2021. Accessed July 4, 2022. https://policyadvice.net/insurance/insights/amazon-alexa-statistics/
- Gagnon MP, Ngangue P, Payne-Gagnon J, Desmartis M. m-Health adoption by healthcare professionals: a systematic review. J Am Med Inform Assoc. 2016;23:212-220.
- NIST. NIST Releases Draft Security Feature Recommendations for IoT Devices. 2019. Accessed July 4, 2022. https://www.nist.gov/news-events/news/2019/08/ nist-releases-draft-security-feature-recommendations-iot-devices
- Office of the National Coordinator for Health Information Technology. What Is FHIR? 2019. Accessed July 4, 2022. https://www.healthit.gov/sites/default/ files/2019-08/ONCFHIRFSWhatIsFHIR.pdf
- Samson LW TW, Turrini G, Sheingold S. Medicare Beneficiaries’ Use of Telehealth in 2020: Trends by Beneficiary Characteristics and Location. 2020. Accessed July 4, 2022. https://www.aspe.hhs.gov/sites/default/files/documents/a1d5d810fe3433e18b192be42dbf2351/medicare-telehealth-report.pdf?_ ga=2.263152908.1288477598.1638811694-1417522139.1637192937
Citation
NG T, B D, S M, TJ R, S B. Practical Applications of the Internet of Things in Radiation Oncology. Appl Radiat Oncol. 2022;(3):7-17.
October 14, 2022