Radiation Recall After the COVID-19 Vaccine: Two Cases and a Review of the Literature
Images
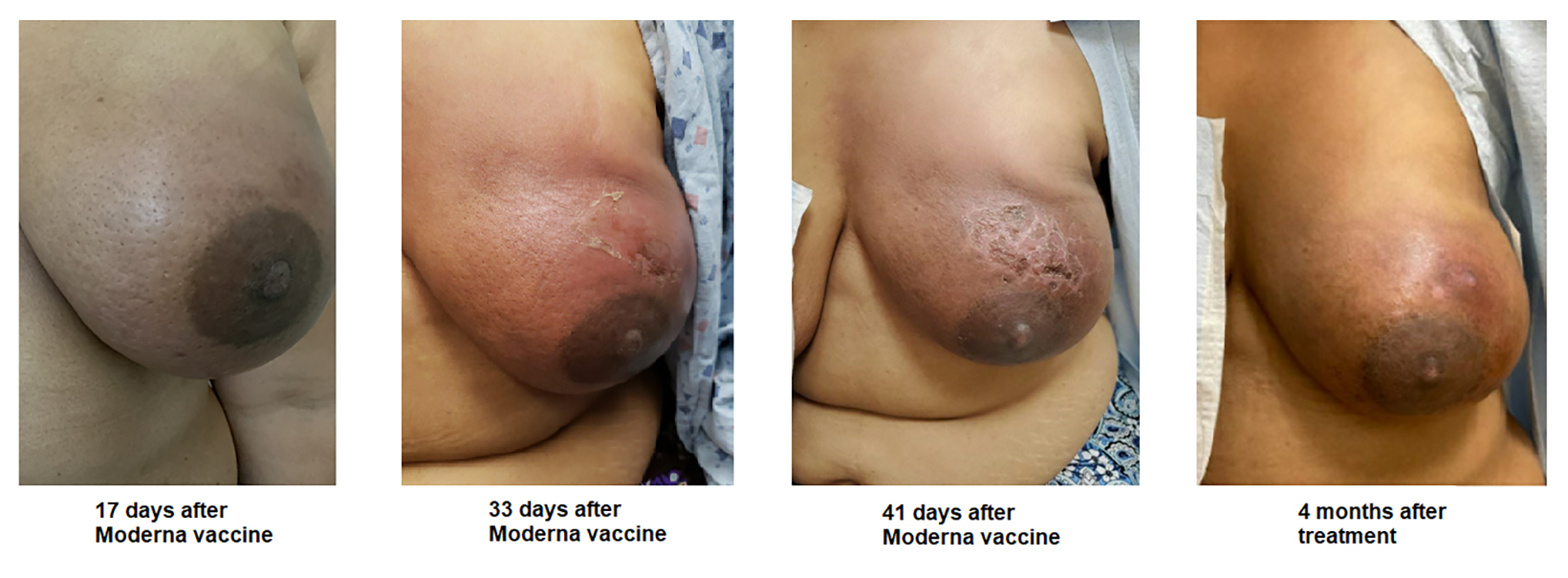
Radiation recall is a localized inflammatory reaction occurring in a previously irradiated site months to years after discontinuing ionizing radiation exposure.1 It is frequently associated with chemotherapy agents, especially anthracyclines. To the best of our knowledge, this report is the first description of radiation recall postmastectomy dermatitis and radiation recall proctitis in 2 patients after the vaccine for COVID-19. Our case series adds to the limited existing literature regarding the potential effects of the vaccine that providers should be aware of when counseling patients.
Case Summary
Case 1
Patient 1 is a 43-year-old woman who initially self-palpated a left breast lump in 2019. She underwent a diagnostic mammogram and ultrasound of her left breast, which showed 2 irregular masses, 1 at the 1 o’clock position 10 cm from the nipple measuring up to 5.8 cm, and 1 at the 2 o’clock position 5 cm from the nipple measuring up to 1.7 cm. She was also found to have abnormal lymph nodes in the left axilla. Core-needle biopsy of her 2 left breast masses and her left axillary lymph node showed invasive ductal carcinoma, grade 3, ER-, PR+, Her2+. Genetic testing was positive for BRCA1 mutation. Metastatic workup including a computed tomography (CT) scan, bone scan, and MRI of the brain was negative. Final stage was cT3 N1 M0.
Patient 1 was started on neoadjuvant docetaxel, carboplatin, Herceptin, and Perjeta (TCHP) for 6 cycles followed by bilateral nipple-sparing mastectomy with left-sided axillary lymph node dissection completed in June 2020. There was minimal residual breast tissue, and the patient had tissue expanders placed at the time of mastectomy for reconstruction. Surgical pathology showed pathologic complete response in the left breast and axilla, ypT0 N0. The patient then started on maintenance Herceptin for 1 year. She also underwent postmastectomy radiation 50 Gy in 25 fractions to the left chest wall and regional lymph nodes (axillary level I-III, supraclavicular [SCV], and internal mammary nodes [IMN]) completed in October 2020. The patient experienced some mild grade 1 dermatitis at the end of treatment, but this fully resolved 1 to 2 months after treatment, with minimal residual hyperpigmentation in the left axilla.
Patient 1 received her first Moderna vaccine in March 2021 and her second on April 2, 2021. On April 12, 2021, the patient developed low- grade fevers and malaise with left breast erythema, edema, and tenderness in the field of prior radiation (Figure 1). This was initially believed to be related to an infection, so the patient was started on Bactrim, with subsequent improvement in erythema and edema. However, skin reaction continued to worsen with development of dry and moist desquamation. She was managed with nitroglycerin cream and Neosporin and was later diagnosed with radiation recall dermatitis. She was also incidentally diagnosed with hypothyroidism by her primary care physician around this time, with TSH of 54, and was started on levothyroxine. After 6 months, the patient’s left breast is nearly fully healed with no more wet desquamation and she will be able to proceed with planned implant exchange surgery. The only persistent clinical finding is some residual hyperpigmented skin, with mild capsular contracture around the tissue expander.
Case 2
Patient 2 is a 69-year-old man who was initially found to have an elevated PSA of 5.1 in 2018. Digital rectal examination showed no palpable masses or nodules (T1c disease). MRI of the prostate showed focal findings suspicious for neoplasm in the left posterolateral peripheral apex with capsular margin intact; overall suspicion was PI-RADS 4/5. Subsequent biopsy showed Gleason 3+4 disease in 3/12 cores. Patient 2 then underwent prostatectomy in October 2018, with surgical pathology showing Gleason 4+3 disease, confined to the prostate without extracapsular or seminal vesicle invasion (pT2), and negative margins. PSA decreased to 0.2 in 2019, but never became undetectable, and later rose to 0.3 in 2020, which was concerning for residual disease. The patient then received 45 Gy in 25 fractions to the pelvic lymph nodes with a sequential boost to 70.2 Gy in 39 fractions to the prostate bed, completed in August 2020. He received concurrent Lupron for 6 months. Patient 2 experienced some mild grade 1 urinary frequency and urgency at the end of treatment, but this fully resolved 2 to 3 months after treatment.
Patient 2 received his first Pfizer vaccine on January 19, 2021, and his second on February 9, 2021. On February 27, 2021, the patient contacted his primary care office reporting worsening rectal bleeding. He then developed severe rectal pain. This prompted a colonoscopy in March, which showed ulcerated friable rectal mucosa in the distal 10 cm of the rectum. Biopsy was consistent with radiation colitis, no dysplasia or malignancy. The patient was treated with Anusol suppositories and Proctofoam. Over the next several months, his rectal ulcer and rectal pain improved and the rectal bleeding resolved. It is unlikely that radiation dose contributed to his proctitis given his rectum V65 was only 18%, much lower than the V65 < 35% acceptable limit.
Discussion
Radiation recall is believed to be a local hypersensitivity reaction triggered by the upregulation of inflammatory cytokines that were already increased in the area of irradiation.1 Reactions typically appear within days to weeks after exposure to the precipitating agent and can vary in severity. The treatment for radiation recall typically involves stopping the precipitating agent and administering steroidal or nonsteroidal anti- inflammatory medications until symptoms resolve.
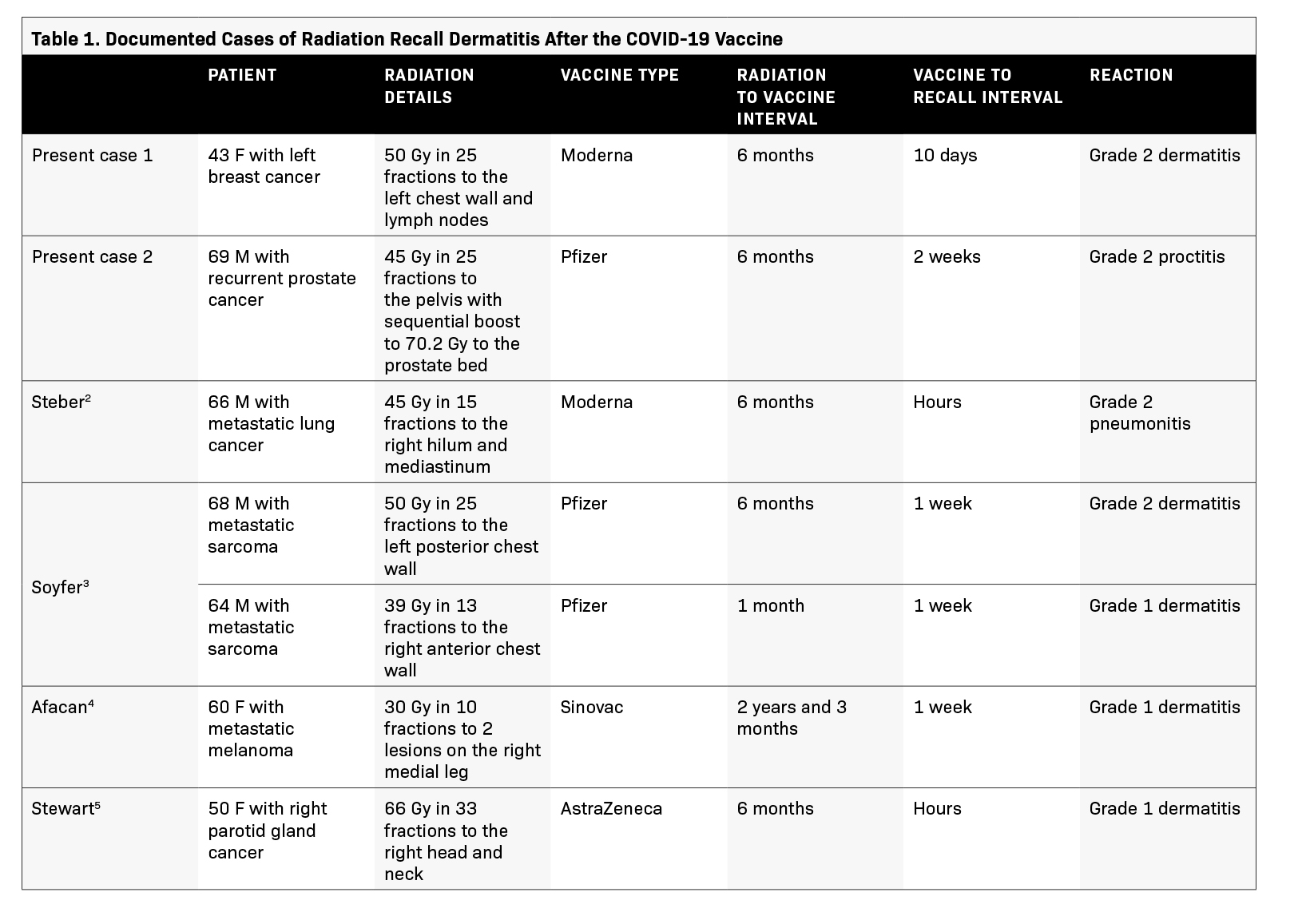
Currently, the incidence of radiation recall after COVID-19 vaccination is unknown, and only 4 published reports have documented such reactions.2-5 A summary of these case reports is in Table 1. Most cases of radiation recall caused by the COVID-19 vaccine are grade 1 to 2 and have mostly occurred in the skin, although there is 1 published case report of a recall pneumonitis occurring in lung.
Radiation recall typically occurred around 5 to 7 days after vaccination, although 2 cases show that it sometimes presents within hours, as described by Stewart et al and Steber et al.2,5 Most patients had a history of prior radiation within 1 year of receiving the COVID-19 vaccine. However, 1 patient experienced radiation recall dermatitis 2 years and 3 months after completion of radiation.4 This was confirmed by biopsy, which showed edema, lymphocyte exocytosis, and dermal collagenization and fibrosis, all consistent with radiation recall dermatitis. It is surprising to see radiation recall phenomenon occur after such a long interval from the time of completion, and more evidence is needed before generalizing. Our 2 patients fit the presentation described in previous case reports.
Initially, it was unclear if patient 1’s newly diagnosed hypothyroidism was related to the postmastectomy radiation or the Moderna vaccine. Hypothyroidism is a known late side effect of curative radiation to the neck region in patients with head and neck cancer and lymphomas, but it is not commonly associated with patients who receive breast irradiation. The incidence of hypothyroidism caused after breast irradiation was characterized by Choi et al.6 It was found that the rate of hypothyroidism at 3 years was 0.8% in patients receiving whole-breast radiation (WBRT) alone and 2.2% in patients receiving regional nodal irradiation, including the SCV (RNI-SCV). The mean thyroid dose was 2 Gy in patients receiving WBRT alone and 8 Gy in patients receiving RNI-SCV. Upon review of patient 1, her mean thyroid dose was around 20 Gy, which likely contributed to her hypothyroidism. This suggests that breast irradiation covering the SCV has a mild to moderate risk of causing hypothyroidism, and providers should consider setting dose constraints to the thyroid gland and counseling patients of this risk. Regarding the risk of hypothyroidism after the COVID-19 vaccine, the incidence is unknown, but most case reports document subacute thyroiditis and not immediate hypothyroidism.7-10 The classic presentation of subacute thyroiditis involves neck pain with initial hyperthyroidism followed by a transient period of hypothyroidism. Patient 1 did not present with neck pain or initial hyperthyroidism, leading us to suspect that the radiation was the causal agent, possibly exacerbated by the patient’s radiation recall dermatitis.
Conclusion
This case report highlights 2 interesting cases of radiation recall, 1 of a patient with prior breast irradiation, and the other with prior pelvic irradiation who developed radiation recall dermatitis and radiation recall proctitis, respectively, within 1 to 2 weeks of receiving the second dose of the COVID-19 vaccine. To the best of our knowledge, this case series is the first description of radiation recall postmastectomy dermatitis and radiation recall proctitis after the COVID-19 vaccine. Both patients had tolerated their course of radiation well, with mild grade 1 acute toxicities, but had a much more significant grade 2 reaction shortly following COVID-19 vaccine administration. It appears that the radiation recall reactions present in many locations of the body, such as skin, lung, and rectum based on published reports. Given the relative novelty of the COVID-19 vaccine, it is important to highlight these 2 cases to better inform providers about the possible effects of the vaccine and help them counsel patients accordingly, especially if the booster vaccine becomes widely available. However, given the rarity of this phenomenon, COVID-19 vaccination should still be highly encouraged for all patients.
References
- Hird AE, Wilson J, Symons S, Sinclair E, Davis M, Chow E. Radiation recall dermatitis: case report and review of the literature. Curr Oncol . 2008;15(1):53-62. doi:10.3747/co.2008.201
- Steber CR, Ponnatapura J, Hughes RT, Farris MK. Rapid development of clinically symptomatic radiation recall pneumonitis immediately following COVID-19 vaccination.
- Cureus, Soyfer V, Gutfeld O, Shamai S, Schlocker A, Merimsky O. COVID-19 vaccine-induced radiation recall phenomenon. Int J Radiat Oncol Biol Phys . 2021;110(4):957-961. doi:10.1016/j.ijrobp.2021.02.048
- Afacan E, Öğüt B, Üstün P, Şentürk E, Yazıcı O, Ad೩şen E. Radiation recall dermatitis triggered by inactivated COVID-19 vaccine. Clin Exp Dermatol . Published online June 2021. doi:10.1111/ced.14786
- Stewart R, McDowell L. Radiation recall phenomenon following COVID-19 vaccination. Int J Radiat Oncol Biol Phys . 2021;111(3):835-836. doi:10.1016/j.ijrobp.2021.06.023
- Choi SH, Chang JS, Byun HK, et al. Risk of hypothyroidism in women after radiation therapy for breast cancer. Int J Radiat Oncol Biol Phys . 2021;110(2):462-472. doi:10.1016/j.ijrobp.2020.12.047
- ೨remli BG, Şendur SN, Ünlütürk U. Three cases of subacute thyroiditis following SARS-CoV-2 vaccine: postvaccination ASIA syndrome. J Clin Endocrinol Metab . 2021;106(9):2600-2605. doi:10.1210/clinem/dgab373
- Franquemont S, Galvez J. Subacute thyroiditis after mRNA vaccine for Covid-19. J Endocr Soc . 2021;5(Supplement_1):A956-A957. doi:10.1210/jendso/bvab048.1954
- Ratnayake GM, Dworakowska D, Grossman AB. Can COVID-19 immunisation cause subacute thyroiditis? Letter to the Editor. Clin Endocrinol (Oxf) . Published online 2021:10-11. doi:10.1111/cen.14555
- Oyibo SO. Subacute thyroiditis after receiving the adenovirus-vectored vaccine for coronavirus disease (COVID-19). Cureus . 2021;13(6):e16045. doi:10.7759/cureus.16045
Citation
J L, J W, C G. Radiation Recall After the COVID-19 Vaccine: Two Cases and a Review of the Literature. Appl Radiat Oncol. 2021;(4):30-33.
December 28, 2021