Study Reveals New Pathway for Brain Tumor Therapy
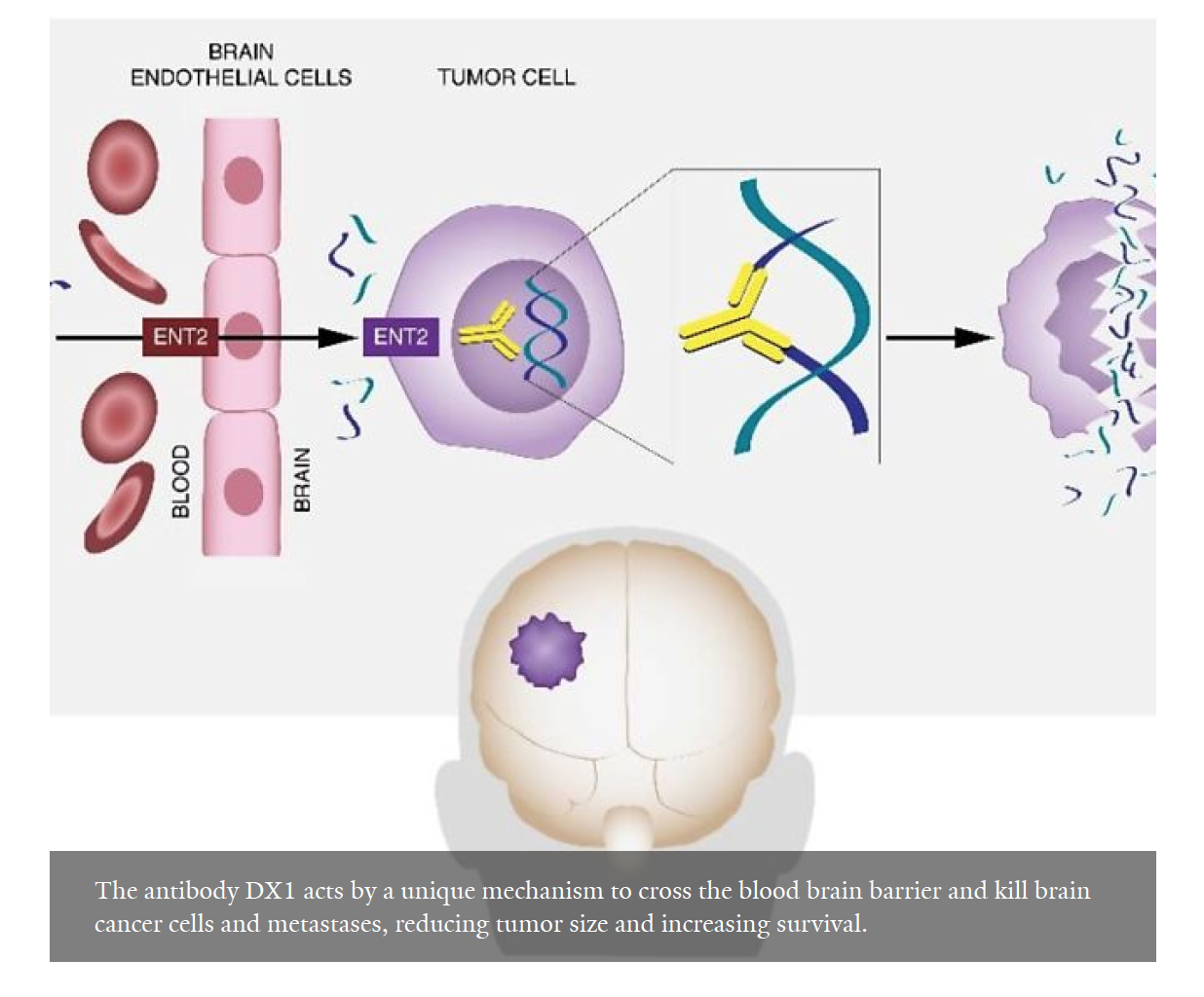 A new Yale Cancer Center study reports the nucleoside transporter ENT2 may offer an unexpected path to circumventing the blood-brain barrier (BBB) and enabling targeted treatment of brain tumors with a cell-penetrating anti-DNA autoantibody.
A new Yale Cancer Center study reports the nucleoside transporter ENT2 may offer an unexpected path to circumventing the blood-brain barrier (BBB) and enabling targeted treatment of brain tumors with a cell-penetrating anti-DNA autoantibody.
“These findings are very encouraging as the BBB prevents most antibodies from penetrating the central nervous system and limits conventional antibody-based approaches to brain tumors,” said James E. Hansen, MD, Associate Professor of Therapeutic Radiology, Radiation Oncology Chief of the Yale Gamma Knife Center at Smilow Cancer Hospital, and corresponding author of the study.
Deoxymab-1 (DX1) is an unusual cell-penetrating autoantibody that localizes into live cell nuclei, inhibits DNA repair, and is synthetically lethal to cancer cells with defects in the DNA damage response (DDR). Researchers have now found that the transporter ENT2 facilitates brain endothelial cell penetration and BBB transport by DX1. In efficacy studies in mice models, DX1 crossed the BBB to suppress orthotopic glioblastoma and breast cancer brain metastases.
“Our data demonstrate the ability of DX1 to cross the BBB and suppress brain tumors in multiple models, and we are particularly impressed that DX1 was able to yield these results as a single agent in these difficult to treat tumor models,” said Jiangbing Zhou, PhD, Associate Professor of Neurosurgery at Yale School of Medicine and co-corresponding author of the study.
“We believe that the ENT2-linked mechanism that transports DX1 across the BBB and into tumors has potential to contribute to multiple new strategies in brain tumor therapy,” added Hansen. “In addition to establishing proof of concept for single agent use of DX1 in brain tumor models, we also now recognize the potential for DX1 to target linked cargo molecules to brain tumors or to be useful as a platform for designing additional brain tumor targeting antibodies, including DX1-based bispecific antibodies.”
Financial support for the study was provided by the National Institute of Neurological Diseases and Stroke of the National Institutes of Health, the Office of the Assistant Secretary of Defense for Health Affairs through the Breast Cancer Research Program, Yale School of Medicine Department of Therapeutic Radiology, Yale Kalimeris and Craig funds, a Lion Heart Fund Pilot Grant, and Patrys Limited, the Australian biotechnology company that has licensed this technology from Yale.