Unexpected Complete Response to Palliative Radiation Therapy in Non-Small Cell Lung Cancer
Applied Radiation Oncology
Published: March 5, 2026
1 U.O.C. Radioterapia Oncologica - Presidio Ospedaliero “Mariano Santo” Contrada Muoio piccolo - Azienda Ospedaliera “Annunziata” di Cosenza, Cosenza, Italy
2 U.O.C. Oncologia Medica - Presidio Ospedaliero “Mariano Santo” Contrada Muoio piccolo - Azienda Ospedaliera “Annunziata” di Cosenza, Cosenza, Italy
3 U.O.S.D. Fisica Medica - Presidio Ospedaliero “Mariano Santo” Contrada Muoio piccolo - Azienda Ospedaliera “Annunziata” di Cosenza, Cosenza, Italy
4 U.O.C. Chirurgia Toracica - Presidio Ospedaliero “Annunziata” - Azienda Ospedaliera “Annunziata” di Cosenza, Cosenza, Italy
5 U.O.C. Medicina Nucleare - Presidio Ospedaliero “Mariano Santo” Contrada Muoio piccolo - Azienda Ospedaliera “Annunziata” di Cosenza, Cosenza, Italy
* Corresponding author: Angelo Andrea Chirillo (chirillo.angelo@gmail.com)
Abstract
Keywords
lung cancer, palliative radiation therapy, pain management, sequential chemo-radiotherapy, tumor down-sizing, unexpected result, multidisciplinary approach, predictive factors, treatment response, case report
Case Summary
A 75-year-old man, a former heavy smoker, presented in October 2023 with a 3-month history of neuropathic chest pain radiating to the right shoulder, associated with progressive functional impairment and a visual analog scale score of 7 despite treatment with paracetamol and codeine. Contrast-enhanced CT identified a large right upper lobe mass with costovertebral infiltration. Staging with PET and MRI excluded distant metastases, consistent with locally advanced non-small cell lung cancer (NSCLC).
The initial therapeutic plan consisted of neoadjuvant chemo radiation therapy; however, due to worsening pain, palliative radiation therapy (8 Gy × 2 fractions, February 13-16, 2024) was administered between the second and third chemotherapy cycles. At completion of chemotherapy (April 2, 2024), the patient exhibited deterioration in performance status and difficulty maintaining treatment position for definitive radiation therapy, prompting referral for surgical management on April 17, 2024. Histopathologic analysis unexpectedly demonstrated a complete pathologic response with no residual neoplastic tissue, representing an extraordinary response to a purely palliative treatment.
Imaging Findings
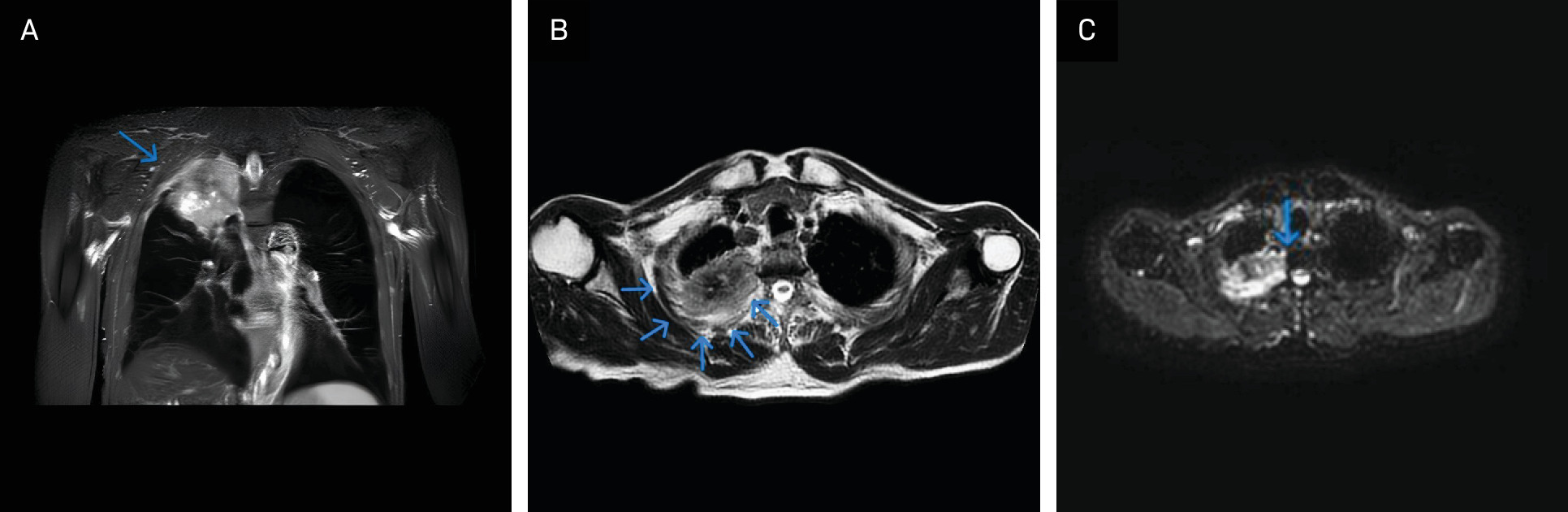
The first relevant imaging evaluation was performed in November 2023 because of suspected Pancoast syndrome. MRI (Figure 1A-C) revealed a mass measuring 7.2 cm (axial) × 6.8 cm (coronal) × 8.9 cm (sagittal) in the right upper lobe, infiltrating the ipsilateral second through 4th ribs and abutting the second through 4th thoracic vertebrae.
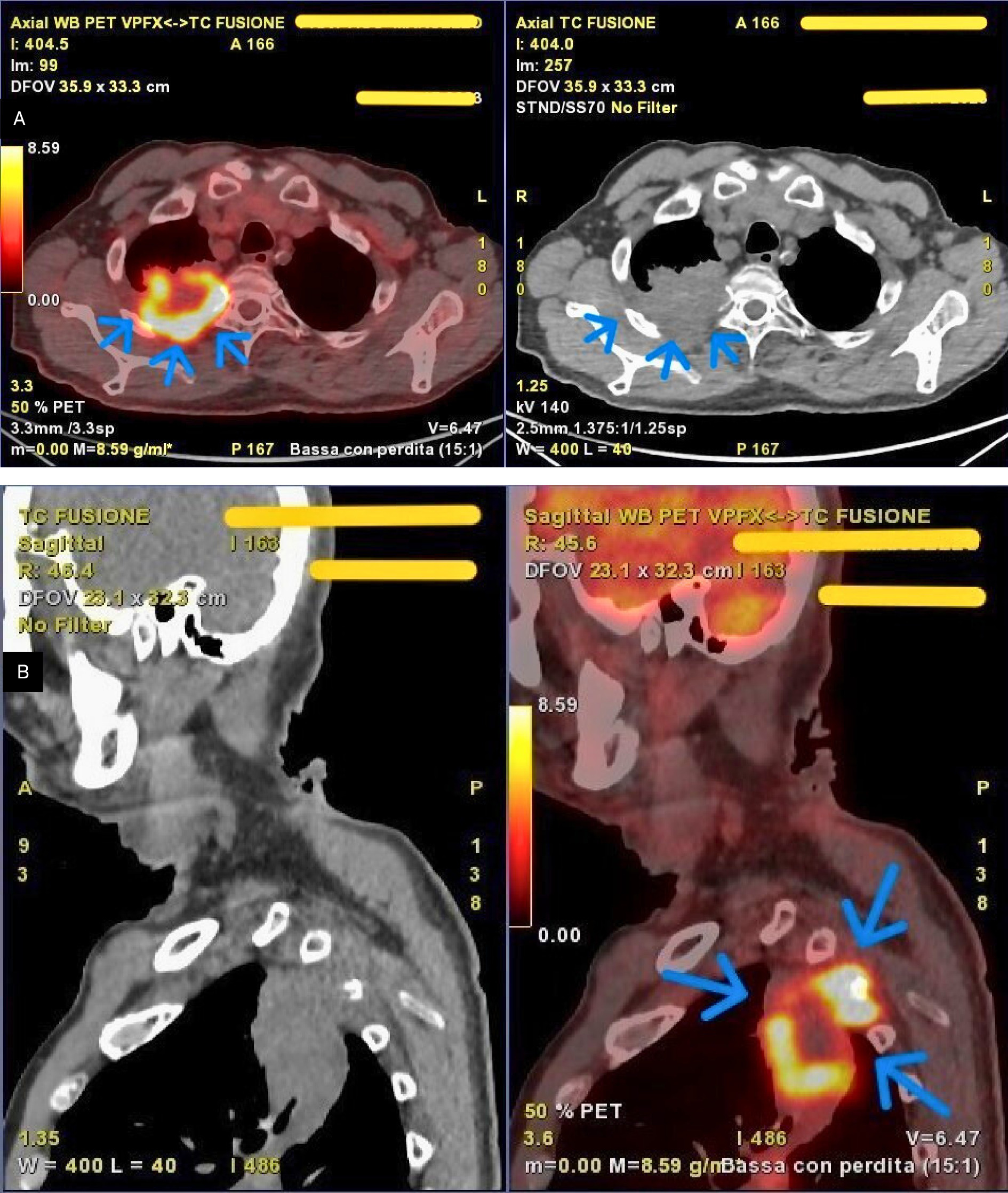
Subsequent whole-body PET (Figure 2A and B) demonstrated intense uptake of fluorodeoxyglucose (SUV_max 19.6) corresponding to the known pulmonary mass, measuring 7.8 cm (axial) × 5.0 cm (coronal) × 9.0 cm (sagittal), located in the apical and posterior segments of the right upper lobe. Moderate tracer uptake suggestive of nodal involvement was observed in the right hilar (SUV_max 3.6) and subcarinal (SUV_max 2.8) regions.
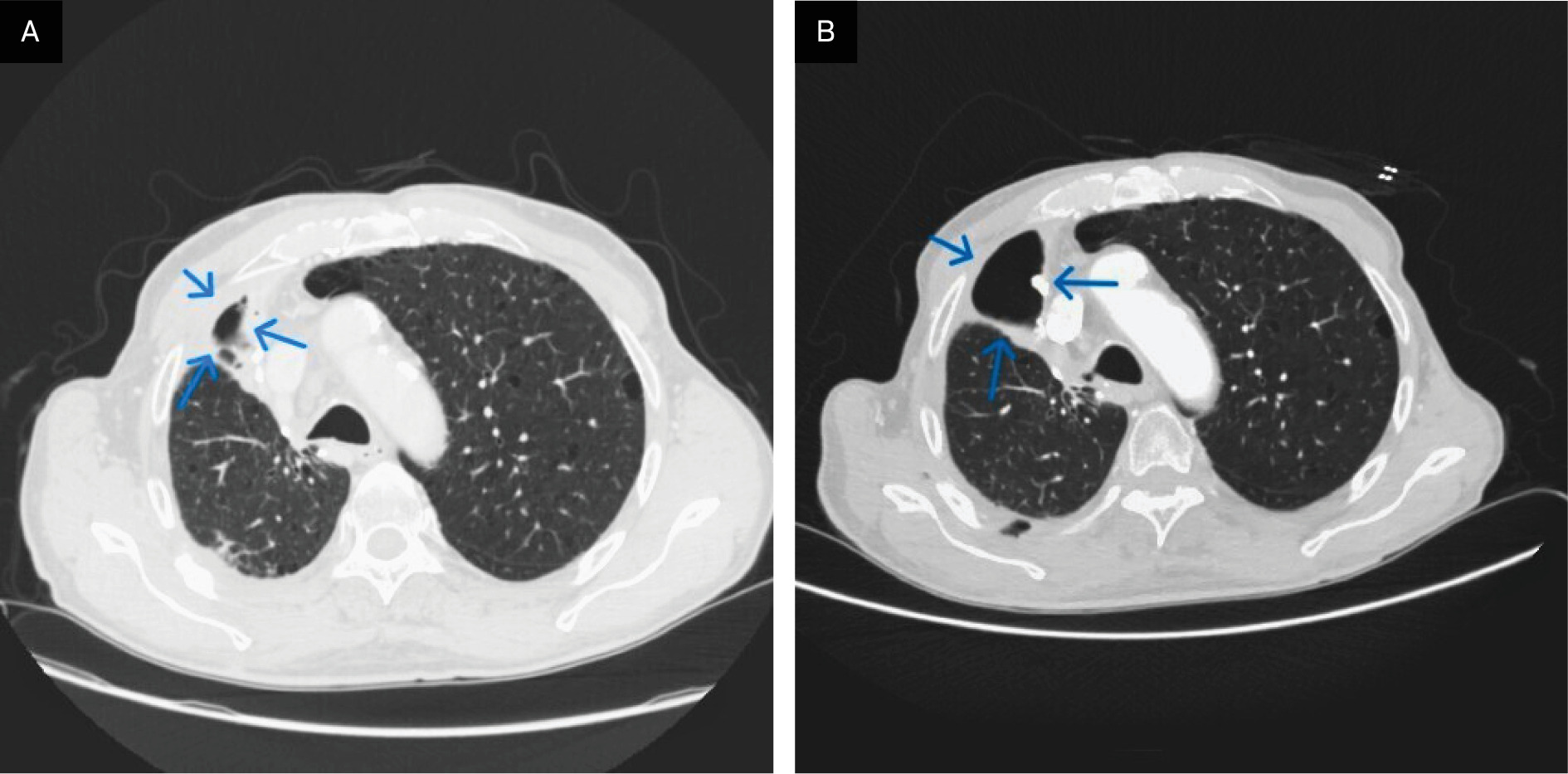
Postoperative thoracic CT (Figure 3A and B) revealed a loculated right-sided hydropneumothorax and confirmed the outcomes of right upper lobectomy with resection of the lateral arches of the second and third ribs. A thin residual pleural effusion and minor subpleural consolidation were present. Paraseptal emphysema persisted in the left upper lobe. Enlarged paratracheal mediastinal lymph nodes, measuring up to 2 cm, were subsequently evaluated histologically by endobronchial US-guided biopsy.
Diagnosis
The patient had a history of hypertension treated with ramipril/hydrochlorothiazide and a previous transient ischemic attack managed with daily acetylsalicylic acid. He had a heavy smoking history (> 20 cigarettes per day for 50 years). He was in good general condition (ECOG performance status 1), with mild weight loss and no dyspnea or hemoptysis. Physical examination revealed reduced mobility of the right upper limb and decreased breath sounds in the right upper lung field. Laboratory tests were within normal limits.
Imaging demonstrated a large right upper lobe mass with infiltration of the second through 4th ribs and the second thoracic vertebra, consistent with advanced disease (cT4 cN2 M0, stage IIIB). Differential diagnoses—including primary bone tumors, soft tissue sarcoma, metastatic disease, plasma cell dyscrasia, and infectious etiologies—were excluded through imaging, laboratory evaluation, and clinical assessment. The diagnosis of locally advanced NSCLC without distant metastases was definitively established by US-guided bronchial biopsy.
Discussion
Although Pancoast tumors account for fewer than 5% of all lung cancer cases, the significant morbidity caused by their infiltrative growth at the thoracic outlet, along with ongoing debate regarding optimal treatment strategies in this anatomically complex region, continues to challenge efforts to improve local control and progression-free survival. 1 These tumors originate from the superior pulmonary sulcus and invade the thoracic inlet, including adjacent ribs and vertebrae. The first clinical and radiologic characterization was described by Pancoast in 1932. 2 Approximately 3 decades later, Paulson introduced a combined approach of preoperative radiation therapy followed by posterior surgical resection, reporting encouraging outcomes. 3
Subsequent therapeutic milestones emerged from pivotal phase II trials in the early 2000s, including SWOG 9416 and JCOG 9806, which demonstrated complete pathologic response rates of 87.3% and 89.4%, respectively, and 5-year overall survival rates of 46% and 61% using combined chemo radiation therapy. 4,5 Technological advancements later enabled dose escalation from the historical 30 to 35 Gy used in earlier protocols to the current standard of 60 Gy, resulting in improved pathologic response and survival outcomes. 6 Radiobiologically, 30 to 35 Gy delivered in 10 fractions corresponds to an EQD2 of 32.5 to 39.4 Gy and a biologically effective dose (BED) of 39 to 47.3 Gy (α/β = 10), whereas a total dose of 60 Gy corresponds to an EQD2 of 60 Gy and a BED of 72 Gy.
In the scientific literature, neoadjuvant treatment strategies for locally advanced NSCLC include either definitive chemo radiation therapy or a trimodality approach incorporating surgery. The principal randomized studies addressing this question are the Intergroup Trial 0139 and the SAKK 16/00 trial. 7,8 In the SAKK 16/00 study, no survival benefit was demonstrated for the trimodality approach using sequential chemo radiation therapy followed by surgery compared with bimodal treatment consisting of chemotherapy and surgery alone. 8 Across published series, reported rates of complete pathologic response range from 15% to 33%, and rates of nodal downstaging (pN0) range from 25% to 46%.
Other investigations have evaluated dose escalation, altered fractionation schedules, and hyperfractionated regimens, demonstrating higher rates of pathologic complete response. However, to date, no studies have shown that a purely palliative radiation therapy regimen, such as 8 Gy × 2 fractions, can induce a complete pathologic response in non–small cell lung cancer. Therefore, the favorable outcome observed in the present case can only be interpreted through hypothesized radiobiologic mechanisms.
Large radiation dose fractions are not limited to direct DNA damage, which represents the classical radiobiologic mechanism. At higher dose levels, radiation also affects tumor vasculature by inducing endothelial apoptosis and ischemic injury through vascular collapse. Tumor endothelial cells are particularly sensitive to high single doses, activating ceramide-mediated signaling pathways that result in apoptosis and disruption of the tumor’s abnormal vasculature. 9,10 Loss of perfusion amplifies direct tumor cell death. Preclinical models have shown that genetic suppression of endothelial apoptosis confers resistance to high-dose radiation, highlighting the therapeutic relevance of doses ≥8 to 10 Gy per fraction. 9,10
High-dose hypofractionated radiation therapy has also been shown to induce immunogenic cell death, characterized by the exposure of damage-associated molecular patterns such as calreticulin and CD47 and the release of ATP and HMGB1. These signals activate dendritic cells and promote cytotoxic T-lymphocyte priming against tumor antigens, thereby enhancing systemic antitumor immunity. 11-14 Preclinical studies suggest that multifraction regimens around 8 Gy (eg, 8 Gy × 3 fractions) may be more immunogenic than single ablative doses. 15-18 Although the regimen used in this case consisted of 8 Gy × 2 fractions, it remains within this biologically active window, potentially allowing vascular, immunologic, and cytotoxic effects to converge.
The exceptional response observed in this patient is likely attributable to the combined effects of direct tumor cell killing, vascular normalization, and immune activation, occurring within a narrow therapeutic window and in an anatomic site where even modest tumor regression can result in substantial clinical benefit, including pain relief and functional recovery.
Conclusion
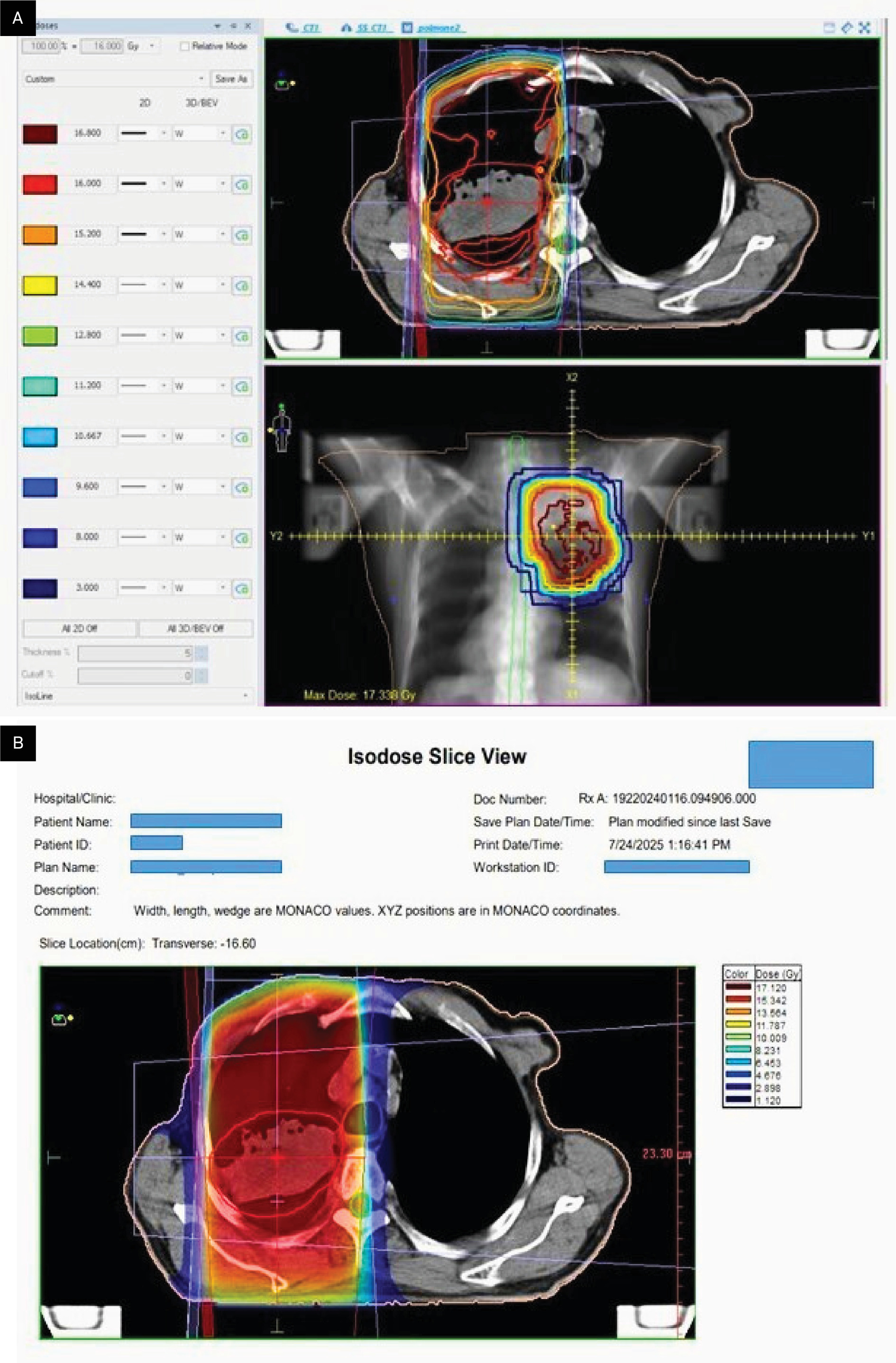
This case represents a remarkable and unexpected complete pathologic response in a patient with superior sulcus NSCLC treated with a short-course palliative radiation therapy regimen of 8 Gy × 2 fractions (Figure 4). This outcome challenges conventional dose–response paradigms and suggests that tumor- and host-specific radiosensitivity factors may play a critical role. Further investigation into ultra-hypofractionated radiation therapy and personalized treatment strategies is warranted.
References
- Foroulis C, Zarogoulidis P, Darwiche K. Superior sulcus (pancoast) tumors: current evidence on diagnosis and radical treatment. J Thorac Dis. 2013;5 Suppl 4(suppl 4):S342-S358. doi:10.3978/j.issn.2072-1439.2013.04.08.
- Pancoast H. Superior pulmonary sulcus tumor: tumor characterized by pain, Horner syndrome, destruction of bone, and atrophy of hand muscles. JAMA. 1932;99:1391-1396. doi:10.1001/jama.1932.02740690001001.
- Shaw R, Paulson D, Kee J. Treatment of superior sulcus tumor by irradiation followed by resection. Ann Surg. 1961;154(1):29-40. doi:10.1097/00000658-196107000-00003.
- Rusch V, Giroux D, Kraut M. Induction chemoradiation and surgical resection for superior sulcus non-small-cell lung carcinomas: long-term results of southwest oncology group trial 9416 (Intergroup trial 0160). J Clin Oncol. 2007;25(3):313-318. doi:10.1200/JCO.2006.08.2826.
- Kunitoh H, Kato H, Tsuboi M. Phase II trial of preoperative chemoradiotherapy followed by surgical resection in patients with superior sulcus non-small-cell lung cancers: report of japan clinical oncology group trial 9806. J Clin Oncol. 2008;26(4):644-649. doi:10.1200/JCO.2007.14.1911.
- Shaw R, Paulson D. Historical radiation dose escalation in superior sulcus tumors: transition from 30–35 gy to contemporary standards of 60 gy. Postgrad Med. 1956;2:147-160. doi:10.1080/00325481.1956.11708264.
- Albain K, Swann R, Rusch V. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet. 2009;374(9687):379-386. doi:10.1016/S0140-6736(09)60737-6.
- Pless M, Stupp R, Ris H. Induction chemoradiation in stage IIIA/N2 non-small-cell lung cancer: a phase 3 randomised trial. Lancet. 2015;386(9998):1049-1056. doi:10.1016/S0140-6736(15)60294-X.
- Garcia-Barros M, Paris F, Cordon-Cardo C. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300(5622):1155-1159. doi:10.1126/science.1082504.
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8(2):89-91. doi:10.1016/j.ccr.2005.07.014.
- Kamarajugadda S, Cai Q, Chen H. Manganese superoxide dismutase promotes anoikis resistance and tumor metastasis. Cell Death Dis. 2013;4(2). doi:10.1038/cddis.2013.20.
- Gameiro S, Jammeh M, Wattenberg M. Radiation-induced immunogenic modulation of tumor enhances antigen processing and calreticulin exposure, resulting in enhanced T-cell killing. Oncotarget. 2014;5(2):403-416. doi:10.18632/oncotarget.1719.
- Galluzzi L, Kepp O, Kroemer G. Immunogenic cell death in radiation therapy. OncoImmunology. 2013;2(10). doi:10.4161/onci.26536.
- Troitskaya O, Novak D, Richter V, Koval O. Immunogenic cell death in cancer therapy. Acta Naturae. 2022;14(1):40-53. doi:10.32607/actanaturae.11523.
- Ko E, Benjamin K, Formenti S. Generating antitumor immunity by targeted radiation therapy: role of dose and fractionation. Adv Radiat Oncol. 2018;3(4):486-493. doi:10.1016/j.adro.2018.08.021.
- Habets T, Oth T, Houben A. Fractionated radiotherapy with 3 × 8 gy induces systemic anti-tumour responses and abscopal tumour inhibition without modulating the humoral anti-tumour response. PLoS ONE. 2016;11(7). doi:10.1371/journal.pone.0159515.
- Craig D, Ambrose S, Stanbery L, Walter A, Nemunaitis J. Systemic benefit of radiation therapy via abscopal effect. Front Oncol. 2022;12. doi:10.3389/fonc.2022.987142.
- Plataniotis G, Theofanopoulou M, Sotiriadou K. Palliative hypofractionated radiotherapy for non–small-cell lung cancer patients previously treated by induction chemotherapy. J Thorac Dis. 2009;1:5-10. doi:10.3978/j.issn.2072-1439.2009.12.01.005.
Disclosures
The authors have no conflicts of interest to disclose. None of the authors received outside funding for the production of this original manuscript and no part of this article has been previously published elsewhere. The patient has provided informed consent for the publication of this case report.
Citation
. Unexpected Complete Response to Palliative Radiation Therapy in Non-Small Cell Lung Cancer. Applied Radiation Oncology. 2026. doi:10.37549/ARO-D-25-0050.