Cardiac-sparing radiation therapy for breast cancer
Images


Radiation therapy has an essential role in the management of breast cancer that includes either postlumpectomy radiation for breast conservation in early stages, or postmastectomy radiation for the chest wall in multiple node-positive or locally advanced stages. Large meta-analyses of prospective randomized trials have confirmed that radiation reduces locoregional recurrences and reduces breast cancer mortality. However, the risk for late cardiac effects caused by the proximity of the heart and coronary vessels to the chest wall or regional nodes has historically mitigated some of these benefits of adjuvant radiation. The Early Breast Cancer Trialists’ Collaborative Group reported a meta-analysis of prospective randomized trials of postmastectomy radiation that noted improved survival in node-positive women.1 The 20-year improvement in breast cancer mortality comparing radiation to no radiation was 8.1% (p = 0.001), but the gain reducing death was only 5.0% (p = 0.01). For women with 1-3 positive nodes, a group in whom the controversy about routine radiation has been particularly intractable, the difference between breast cancer mortality and survival was 7.9% (p = 0.01) and 3% (p = ns).
This 3% to 5% difference between breast cancer mortality and overall survival may, in large part, be due to an excess of cardiac disease caused by the radiation of that era of studies in the meta-analysis from 1964 to 1986.1 In the Surveillance Epidemiology and End Results (SEER) database from 1973-1992, there was an excess rate of fatal myocardial infarction of 1% to 2% over the course of 8 to 18 years from treatment for patients receiving left-sided vs. right-sided adjuvant radiation.2 A loss of 1% was also seen between the improvement in breast cancer mortality and overall survival in the postlumpectomy radiation setting from randomized prospective trials conducted between 1976-1999.3 This greater difference of death from nonbreast cancer causes between the postmastectomy and postlumpectomy trials may be decreasing over decades due in part to technical improvements, but the difference may also be due to the greater use of regional node — specifically internal mammary node — radiation in the earlier postmastectomy trials. Radiation of the chest wall and internal mammary nodes (IMNs) has been specifically linked to coronary stenosis in distributions consistent with the radiation fields of conventional radiation.4 For fear of late cardiac injury if IMNs were included for left-sided breast cancer patients, a large prospective population-based cohort study of internal mammary node irradiation treated right-sided patients only.5
In a retrospective review of 2,168 women treated for breast cancer from 1958 to 2001, heart dose was estimated from idealized phantom measurements.6 They found that the mean heart dose correlated with excess relative risk of coronary events by 7.4% per 100 cGy. In that period, the mean heart dose was estimated to be 6.6 Gy for women with tumors in the left breast. In a systematic review of 149 studies published during 2003 to 2013, the mean heart dose from left-sided breast radiation therapy was 5.4 Gy.7 The lowest mean heart doses were from tangential radiation with breathing control (1.3 Gy) or proton radiation (0.5 Gy), and the highest inclusion of internal mammary lymph nodes (8 Gy). Aiming to reduce the mean dose is an important goal for modern radiation therapy in order to reduce ultimate late cardiac complications. In this way, the survival improvements associated with adjuvant radiation could be further improved if excess cardiac deaths could be eliminated altogether.
Forward Planning
Early whole-breast irradiation used photon beam 2D techniques consisting of opposed tangential beams of uniform radiation intensity across the field that could be modified with wedge compensators. The introduction of 3-dimensional computed tomography (3D CT) planning in the 1990s permitted the calculation of heart dose in a more precise manner than just observing the amount of the heart silhouette in a tangential portal film. 8 Early attempts to limit heart dose in a breast tangent would be adjusting the beam angle to avoid the heart or adding a block over the heart silhouette.9 Forward planning then developed to optimize dose heterogeneity within the target by manually creating smaller fields using custom blocking or multileaf collimation within a larger tangent — what is known as a “field-in-field” technique.10 In early experiences, such techniques of using beams of nonuniform fluence applied to a target structure were labeled as intensity-modulated radiation therapy (IMRT) but today are considered and reimbursed as 3D conformal radiation. Forward-planned tangential radiation has been shown to be superior to 2D tangential radiation using wedges in 3 prospective randomized trials for reducing desquamation, late skin telangeictasias and fibrosis.11-13 The 3D conformal tangents with forward planning with custom blocking or predefined segments can decrease the heart dose14,15 and normal tissue complication probability for late cardiac toxicity on average by 30%16 compared to using simple wedged tangents.
Prone Positioning
Prone positioning may have advantages for some women with large or pendulous breasts, or left-sided breast cancers compared to traditional supine positioning. When supine, large- or pendulous-breasted women often have a large separation, or width, between the posterior entry and exit points of the tangential radiation field. This is a cause for large dose inhomogeneity that may only be partially overcome by advances in 3D conformal or IMRT. These women may also have large skin folds particularly in the inframammary region that increase acute dermatitis and risk for moist desquamation. For left-sided women, the lateral displacement of the breast in large women may require a deeper tangent for breast coverage that increases heart dose.
Prone positioning can reduce chest wall separation, deep skin folds, dose inhomogeneity, and heart dose for a large majority of breast cancer patients.17,18 Prone is generally limited to treatment of the breast only, or breast and low axilla,19 but full regional nodal coverage of the high axilla, supraclavicular and internal mammary nodes is generally not possible in the prone position. In addition, caution is needed during simulation for patient selection — judgment of the cardiac anatomy and possible breast tangent — because in a small minority of patients, prone positioning may increase heart dose. In a comparison study of 30 left-sided patients simulated both prone and supine, prone positioning reduced heart and left anterior descending (LAD) doses in 19 patients, increased it in 8 patients, and had no effect in 3 patients.20 In a prospective study of 200 left-sided patients simulated both supine and prone, prone position was associated with an 85% reduction of in-field heart volumes compared to supine.21 This did not reach significance in small-breasted women. A benefit was seen in 85% of patients to prone positioning for the heart volume in the radiation field, but supine position was better for 15%.
Intensity-modulated Radiation Therapy
IMRT describes an inverse planning technique in which beams of nonuniform fluence are created by optimizing coverage of a planning target volume (PTV). Much use of IMRT in adjuvant treatment of breast cancer has been using standard tangential beam arrangements. A benefit in dose homogeneity with inverse planned or hybrid IMRT techniques compared to forward-planned 3D conformal has been shown in some studies22,23 but not all.24 IMRT has been reported to reduce dose to heart compared to 3D in most studies25-27 but not others.28 There can be significant variation in patient anatomy so that there are overlapping ranges of heart dose for IMRT vs. 3D, and IMRT may be superior to 3D in heart dose for some patients but not all.14 There may also be a tradeoff in reduced PTV coverage with IMRT that prioritizes cardiac sparing.14,15 In some studies, an added benefit for IMRT is an overall reduced planning time and decreased dependence on dosimetrist experience compared to 3D conformal.29,30
ASTRO’s Choosing Wisely campaign advocated against the routine use of IMRT to deliver whole-breast radiation therapy. The randomized Canadian multicenter study that showed reduced acute toxicity from tangential radiation with IMRT compared to 2D tangents did include patients treated with forward-planned or inverse-planned IMRT.11 However, the Cambridge Breast IMRT trial did not show a reduction in toxicity.31 The rates of IMRT for breast cancer increased dramatically from 2001 to 2011,32,33 and this increase in IMRT usage is associated with a markedly higher cost for adjuvant radiation.32 The Radiation Therapy Oncology Group trial 1005 was a phase III trial that created a database of CT plans for approximately 2,000 patients treated with whole-breast radiation from 2011 to 2014. The trial allowed field-in-field 3D conformal or IMRT as long as preset minimum constraints could be met. A subgroup analysis of differences in mean heart dose and late toxicity outcomes will be a useful prospective, although not randomized, comparison.
Certain patient subgroups may benefit from inverse planning IMRT compared to 3D conformal. This could be an option for some cases of challenging anatomy, such as large chest wall separation causing dose inhomogeneity; left-sided cases with a large amount of heart close to the chest wall or pectus excavatum; or where internal mammary node irradiation is needed. Inverse-planned IMRT has been shown to improve dosimetric coverage, homogeneity, and high doses received by the lung and heart for patients requiring internal mammary node irradiation compared to partly wide tangents or mixed-beam plans. 26 However, the tradeoff is that the addition of nontangential beams to IMRT increases the low-dose radiation to the heart and V5 dose.34-38 IMRT should be considered and comparison plans created when 3D conformal forward planning is not able to achieve the initial desired dose goals.
Respiratory Control
There are several commercially available methods for respiratory control during radiation therapy for breast cancer. The purpose is to use an increase in lung volume and inferior displacement of the diaphragm to increase the distance between the heart and the breast/chest wall to reduce radiation dose. In one method, an active-breathing control (ABC) device is used for regulation of respiratory inspiratory volume. The other method relies on patient coaching for voluntary deep inspiration breath holding (DIBH) that is verified with either direct volume measurement or surface anatomy verification.
Studies comparing mean heart dose with free breathing vs. respiratory control are shown in Table 1. In one study, moderate DIBH with ABC in 87 of 99 (88%) patients was associated with a mean heart dose of 254 cGy compared to 423 cGy with free breathing (FB) (p < 0.001).39 In a prospective study of ABC for left-sided breast cancer, 72% of enrolled patients were ultimately treated with ABC with inability to tolerate the procedure being the predominant cause for ineligibility.40 The mean heart dose was reduced by ABC compared to FB by > 20% in 88% of patients, and the median mean heart dose was 270 cGy for FB compared with 90 cGy for ABC. Mast et al compared free breathing (FB) to DIBH plans with tangential 3D conformal and IMRT techniques.27 For the heart and LAD-region, a significant dose reduction was found with DIBH (p < 0.01). The mean heart dose for 3D vs. IMRT in 20 patients was 180 cGy compared to 150 cGy in DIBH, and 330 cGy and 270 cGy in FB, respectively (p = 0.01). In a prospective study of 17 left-sided patients, supine position with DIBH significantly reduced the volume of the heart receiving 30 Gy, the mean heart dose, and mean LAD coronary artery dose compared to supine with FB and prone positioning.41 In a study of 35 patients planned with FB or DIBH, mean dose for heart was 90 cGy vs. 250 cGy, (p < 0.0001)42 and in 75% of patients there was felt to be a benefit to DIBH. In a prospective registry of 150 patients, in which patients were selected for FB (38) or DIBH (110) at physician discretion, DIBH plans were associated with a mean heart dose of 137.6 cGy compared to 255.7 cGy with FB (p < 0.0001).43 On multivariate analysis, younger age, higher BMI, and larger change in lung volume between scans were associated with a greater change in mean heart dose between techniques.
The improvement of cardiac dose with respiratory control now seems well settled. These techniques have been shown to be clinically practical and have no significant impact on patient treatment time and throughput.43,44 Whether this will lead to clinically evident reduction in cardiac events is unknown. In one prospective study of ABC vs. FB, there was decreased dose to the left ventricle but no change in myocardial perfusion changes 6 months after treatment.45 Further research is also needed to determine how best to select patients. The IMN chain may be particularly sensitive to changes in position and dose coverage with respiratory motion,46,47 and ABC has been shown to improve heart dose, particularly in the setting of IMN irradiation.48 All patients with need for internal mammary node radiation would seem good candidates for respiratory control. However, treating all left-sided patients who may tolerate it may also lead to overutilization of resources in a significant minority of patients who may be appropriately treated with FB. Further research is needed to determine whether physicians can appropriately select patients at the time of simulation on a case-by-case basis,43 or whether objective measures may predict accurately who will benefit most from respiratory control.42
Proton Beam Radiation
Proton radiation therapy may have dosimetric advantages compared to photons due to the property of the positively charged proton depositing the bulk of its energy in tissue in a finite range, or Bragg peak, with essentially no residual radiation beyond this depth. In clinical application to breast cancer, this could theoretically allow full breast or nodal target coverage within the Bragg peak with no dose to heart and lung posteriorly beyond the Bragg peak. Dosimetric studies have demonstrated the superiority of proton therapy in the postmastectomy radiation therapy setting with respect to low doses to organs-at-risk while maintaining superior target coverage, particularly regional nodes.49,50 In a report of 12 patients treated in a prospective clinical trial, 11 left-sided patients achieved an average mean heart dose of 44 cGy, and had 75% grade 2 acute skin toxicity (no grade 3) and only 1 grade 3 toxicity (fatigue).51 In a report of 30 patients, most treated to internal mammary nodes, the mean heart dose achieved was 1 Gy for left-sided patients.52 There was grade 2 dermatitis in 71%, moist desquamation in 29%, grade 2 esophagitis in 29%, and 1 grade 3 reconstructive complication. Proton therapy may reduce risk for cardiac toxicity of radiation compared to photon radiation by not only reducing mean heart dose, but dose to the critical coronary artery structures on the heart’s surface.53 In one study, a scanning proton technique for left-sided irradiation was associated with lower minimum, maximum, and dose to 0.2 cc of the LAD coronary artery than the best possible photon beam radiation technique (IMRT with DIBH).54
In practice, there are several limita-tions of protons. Coverage of the width of the breast and other targets in the patient requires creation of a wider spread-out Bragg peak (SOBP) that increases skin dose. Proton therapy distal range has intrinsic uncertainty that can lead to overshooting or undershooting the posterior target edge, and greater sensitivity to patient or organ motion. The potential advantage to protons is thought to be physical and not biological — protons are estimated to have a relative biologic effectiveness (RBE) of 1.1 compared to photons, which is taken into account for dose calculations by treatment planning systems. In actuality, there may be variation of proton linear energy transfer along the track length causing lower RBE in the SOBP and higher RBE at the end track that could potentially lower tumor control or increase complication probabilities compared to current planning system estimates.55 Current methods of proton techniques such as double scattering have limitations in field size, matching, and dose shaping. More advanced techniques like pencil-beam scanning and intensity-modulated proton therapy could potentially treat some of the most challenging postmastectomy radiation therapy cases, due to breast reconstruction, internal mammary node coverage, or lower skin dose, but may not be clinically deliverable with current equipment.53,56
The RADCOMP breast proton vs. photon study [NCT02603341] is being conducted on the hypothesis that proton therapy for locally advanced breast cancer reduces major cardiovascular events, is noninferior in cancer control, and improves health-related quality of life compared to photon therapy. Participants in the trial will be randomized to either proton or photon therapy. The inclusion criteria is broad: mastectomy with or without reconstruction or lumpectomy, any type of axillary surgery, any adjuvant or neoadjuvant chemotherapy, and left- or right-sided breast cancer as long as internal mammary nodes are intended to be treated.
Conclusion
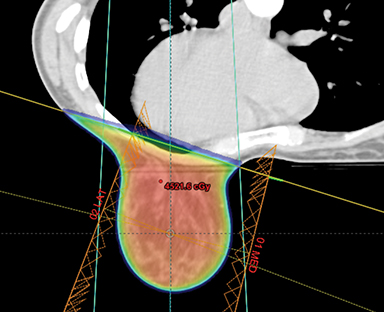
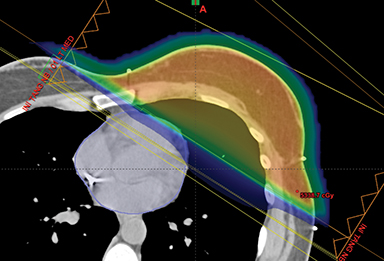
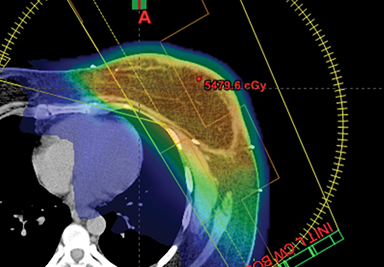
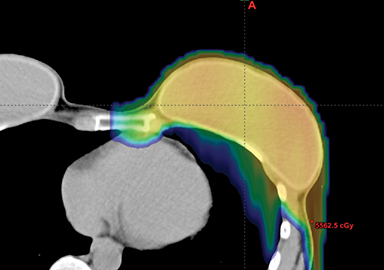
This report has reviewed the wide variety of techniques for adjuvant breast or chest wall radiation therapy for minimizing heart dose. Field-in-field 3D conformal (forward planning) may be seen as the current minimum standard for breast patients today (Figure 1). In many cases, greater cardiac sparing can be achieved with prone positioning (Figure 2), DIBH (Figure 3), IMRT with 2 or more fixed angles (Figure 3), IMRT with arcs (Figure 4), or protons (Figure 5). One challenge to the practicing clinician is acquiring the equipment and experience to have one or more of the options available for their patients, which is subject to constraints on department staff and resources. In a large radiation therapy department with all of these potential options, or a region where referral to specialty centers is possible, another challenge is developing the experience to select patients a priori or at the time of simulation for one or the other modality. Matching the best approach for each patient’s unique target needs and anatomy is necessary instead of a one-size-fits-all approach to cardiac avoidance.
References
- Early Breast Cancer Trialists’ Collaborative Group, McGale P, Taylor C, et al. Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet. 2014;383:2127-2135.
- Paszat LF, Mackillop WJ, Groome PA, et al. Mortality from myocardial infarction after adjuvant radiotherapy for breast cancer in the Surveillance, Epidemiology, and End-Results cancer registries. J Clin Oncol. 1998;16:2625-2631.
- Early Breast Cancer Trialists’ Collaborative Group, Darby S, McGale P, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet. 2011;378:1707-1716.
- Nilsson G, Holmberg L, Garmo H, et al. Distribution of coronary artery stenosis after radiation for breast cancer. J Clin Oncol. 2012;30:380-386.
- Thorsen LB, Offersen BV, Dano H, et al. DBCG-IMN: A population-based cohort study on the effect of internal mammary node irradiation in early node-positive breast cancer. J Clin Oncol. 2016;34:314-320.
- Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med. 2013;368:987-998.
- Taylor CW, Wang Z, Macaulay E, et al. Exposure of the heart in breast cancer radiation therapy: a systematic review of heart doses published during 2003 to 2013. Int J Radiat Oncol Biol Phys. 2015;93:845-853.
- Das IJ, Cheng EC, Freedman G, Fowble B. Lung and heart dose volume analyses with CT simulator in tangential field irradiation of breast cancer. Int J Radiat Oncol Biol Phys. 1998;42:11-19.
- Raj KA, Evans ES, Prosnitz RG, et al. Is there an increased risk of local recurrence under the heart block in patients with left-sided breast cancer? Cancer J. 2006;12:309-317.
- Vicini FA, Sharpe M, Kestin L, et al. Optimizing breast cancer treatment efficacy with intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2002;54:1336-1344.
- Pignol JP, Olivotto I, Rakovitch E, et al. A multicenter randomized trial of breast intensity-modulated radiation therapy to reduce acute radiation dermatitis. J Clin Oncol. 2008;26:2085-2092.
- Donovan E, Bleakley N, Denholm E, et al. Randomised trial of standard 2D radiotherapy (RT) versus intensity modulated radiotherapy (IMRT) in patients prescribed breast radiotherapy. Radiother Oncol. 2007;82:254-264.
- Mukesh MB, Barnett GC, Wilkinson JS, et al. Randomized controlled trial of intensity-modulated radiotherapy for early breast cancer: 5-year results confirm superior overall cosmesis. J Clin Oncol. 2013;31:4488-4495.
- Landau D, Adams EJ, Webb S, Ross G. Cardiac avoidance in breast radiotherapy: a comparison of simple shielding techniques with intensity-modulated radiotherapy. Radiother Oncol. 2001;60:247-255.
- Cho BC, Schwarz M, Mijnheer BJ, Bartelink H. Simplified intensity-modulated radiotherapy using pre-defined segments to reduce cardiac complications in left-sided breast cancer. Radiother Oncol. 2004;70:231-241.
- Hurkmans CW, Cho BCJ, Damen E, et al. Reduction of cardiac and lung complication probabilities after breast irradiation using conformal radiotherapy with or without intensity modulation. Radiother Oncol. 2002;62:163-171.
- Merchant TE, McCormick B. Prone position breast irradiation. Int J Radiat Oncol Biol Phys. 1994;30:197-203.
- Mahe MA, Classe JM, Dravet F, et al. Preliminary results for prone-position breast irradiation. Int J Radiat Oncol Biol Phys. 2002;52:156-160.
- MacDermed DM, Houtman KM, Thang SH, et al. Therapeutic radiation dose delivered to the low axilla during whole breast radiation therapy in the prone position: implications for targeting the undissected axilla. Pract Radiat Oncol. 2014;4:116-122.
- Kirby AM, Evans PM, Donovan EM, et al. Prone versus supine positioning for whole and partial-breast radiotherapy: a comparison of non-target tissue dosimetry. Radiother Oncol. 2010;96:178-184.
- Formenti SC, DeWyngaert JK, Jozsef G, Goldberg JD. Prone vs supine positioning for breast cancer radiotherapy. JAMA. 2012;308:861-863.
- Mihai A, Rakovitch E, Sixel K, et al. Inverse vs. forward breast IMRT planning. Med Dosim. 2005;30: 149-154.
- Descovich M, Fowble B, Bevan A, et al. Comparison between hybrid direct aperture optimized intensity-modulated radiotherapy and forward planning intensity-modulated radiotherapy for whole breast irradiation. Int J Radiat Oncol Biol Phys. 2010;76: 91-99.
- Mayo CS, Urie MM, Fitzgerald TJ. Hybrid IMRT plans—concurrently treating conventional and IMRT beams for improved breast irradiation and reduced planning time. Int J Radiat Oncol Biol Phys. 2005;61:922-932.
- Coon AB, Dickler A, Kirk MC, et al. Tomotherapy and multifield intensity-modulated radiotherapy planning reduce cardiac doses in left-sided breast cancer patients with unfavorable cardiac anatomy. Int J Radiat Oncol Biol Phys. 2010;78:104-110.
- Beckham WA, Popescu CC, Patenaude VV, et al. Is multibeam IMRT better than standard treatment for patients with left-sided breast cancer? Int J Radiat Oncol Biol Phys. 2007;69:918-924.
- Mast ME, van Kempen-Harteveld L, Heijenbrok MW, et al. Left-sided breast cancer radiotherapy with and without breath-hold: does IMRT reduce the cardiac dose even further? Radiother Oncol. 2013;108: 248-253.
- Taunk NK, Prosnitz RG. Planning comparison of intensity modulated radiation therapy delivered with 2 tangential fields versus 3-dimensional conformal radiotherapy for cardiac sparing in women with left-sided breast cancer. Pract Radiat Oncol. 2012;2:248-256.
- Descovich M, Fowble B, Bevan A. Comparison between hybrid direct aperature optimized intensity- modulated radiotherapy and forward planning intensity-modulated radiotherapy for whole breast irradiation. Int J Radiat Oncol Biol Phys. 2010;76:91-99.
- Mayo CS, Urie MM, Fitzgerald TJ. Hybrid IMRT plans-concurrently treating conventional and IMRT beams for improved breast irradiation and reduced planning time. Int J Radiat Oncol Biol Phys. 2005;61:922-932.
- Barnett GC, Wilkinson J, Moody AM, et al. A randomised controlled trial of forward-planned radiotherapy (IMRT) for early breast cancer: baseline characteristics and dosimetry results.Radiother Oncol. 2009;92:34-41.
- Smith BD, Pan IW, Shih YC, et al. Adoption of intensity-modulated radiation therapy for breast cancer in the United States. J Natl Cancer Inst. 2011;103:798-809.
- Wang EH, Mougalian SS, Soulos PR, et al. Adoption of intensity modulated radiation therapy for early-stage breast cancer from 2004 through 2011. Int J Radiat Oncol Biol Phys. 2015;91:303-311.
- Farace P, Zucca S, Solla I, et al. Planning hybrid intensity modulated radiation therapy for whole-breast irradiation. Int J Radiat Oncol Biol Phys. 2012;84:e115-122.
- Schubert LK, Gondi V, Sengbusch E, et al. Dosimetric comparison of left-sided whole breast irradiation with 3DCRT, forward-planned IMRT, inverse-planned IMRT, helical tomotherapy, and topotherapy. Radiother Oncol. 2011;100:241-246.
- Viren T, Heikkila J, Myllyoja K, et al. Tangential volumetric modulated arc therapy technique for left-sided breast cancer radiotherapy. Radiat Oncol. 2015;10:79.
- Popescu CC, Olivotto IA, Beckham WA, et al. Volumetric modulated arc therapy improves dosimetry and reduces treatment time compared to conventional intensity-modulated radiotherapy for locoregional radiotherapy of left-sided breast cancer and internal mammary nodes. Int J Radiat Oncol Biol Phys. 2010;76:287-295.
- Jagsi R, Moran J, Marsh R, et al. Evaluation of four techniques using intensity-modulated radiation therapy for comprehensive locoregional irradiation of breast cancer. Int J Radiat Oncol Biol Phys. 2010;78:1594-1603.
- Swanson T, Grills IS, Ye H, et al. Six-year experience routinely using moderate deep inspiration breath-hold for the reduction of cardiac dose in left-sided breast irradiation for patients with early-stage or locally advanced breast cancer. Am J Clin Oncol. 2013;36:24-30.
- Eldredge-Hindy H, Lockamy V, Crawford A, et al. Active breathing coordinator reduces radiation dose to the heart and preserves local control in patients with left breast cancer: report of a prospective trial. Pract Radiat Oncol. 2015;5:4-10.
- Verhoeven K, Sweldens C, Petillion S, et al. Breathing adapted radiation therapy in comparison with prone position to reduce the doses to the heart, left anterior descending coronary artery, and contralateral breast in whole breast radiation therapy. Pract Radiat Oncol. 2014;4:123-129.
- Rochet N, Drake JI, Harrington K, et al. Deep inspiration breath-hold technique in left-sided breast cancer radiation therapy: Evaluating cardiac contact distance as a predictor of cardiac exposure for patient selection. Pract Radiat Oncol. 2015;5:e127-134.
- Tanguturi SK, Lyatskaya Y, Chen Y, et al. Prospective assessment of deep inspiration breath-hold using 3-dimensional surface tracking for irradiation of left-sided breast cancer. Pract Radiat Oncol. 2015;5:358-365.
- Comsa D, Barnett E, Le K, et al. Introduction of moderate deep inspiration breath hold for radiation therapy of left breast: Initial experience of a regional cancer center. Pract Radiat Oncol. 2014;4: 298-305.
- Zellars R, Bravo PE, Tryggestad E, et al. SPECT analysis of cardiac perfusion changes after whole-breast/chest wall radiation therapy with or without active breathing coordinator: results of a randomized phase 3 trial. Int J Radiat Oncol Biol Phys. 2014;88:778-785.
- Frazier RC, Vicini FA, Sharpe MB, et al. Impact of breathing motion on whole breast radiotherapy: a dosimetric analysis using active breathing control. Int J Radiat Oncol Biol Phys. 2004;58:1041-1047.
- Qi XS, White J, Rabinovitch R, et al. Respiratory organ motion and dosimetric impact on breast and nodal irradiation. Int J Radiat Oncol Biol Phys. 2010;78:609-617.
- Remouchamps VM, Vicini FA, Sharpe MB, et al. Significant reductions in heart and lung doses using deep inspiration breath hold with active breathing control and intensity-modulated radiation therapy for patients treated with locoregional breast irradiation. Int J Radiat Oncol Biol Phys. 2003;55:392-406.
- Ares C, Khan S, Macartain AM, et al. Postoperative proton radiotherapy for localized and locoregional breast cancer: potential for clinically relevant improvements? Int J Radiat Oncol Biol Phys. 2010;76:685-697.
- Xu N, Ho MW, Li Z. Can proton therapy improve the therapeutic ratio in breast cancer patients at risk for nodal disease? Am J Clin Oncol. 2014;37:568-574.
- MacDonald SM, Patel SA, Hickey S, et al. Proton therapy for breast cancer after mastectomy: early outcomes of a prospective clinical trial. Int J Radiat Oncol Biol Phys. 2013;86:484-490.
- Cuaron JJ, Chon B, Tsai H, et al. Early toxicity in patients treated with postoperative proton therapy for locally advanced breast cancer. Int J Radiat Oncol Biol Phys. 2015;92:284-291.
- Mast ME, Vredeveld EJ, Credoe HM, et al. Whole breast proton irradiation for maximal reduction of heart dose in breast cancer patients. Breast Cancer Res Treat. 2014;148:33-39.
- Lin LL, Vennarini S, Dimofte A, et al. Proton beam versus photon beam dose to the heart and left anterior descending artery for left-sided breast cancer. Acta Oncol. 2015;54:1032-1039.
- Carabe A, Espana S, Grassberger C, Paganetti H. Clinical consequences of relative biological effectiveness variations in proton radiotherapy of the prostate, brain and liver. Phys Med Biol. 2013;58:2103-2117.
- Jimenez RB, Goma C, Nyamwanda J, et al. Intensity modulated proton therapy for postmastectomy radiation of bilateral implant reconstructed breasts: a treatment planning study. Radiother Oncol. 2013;107:213-217.
Citation
Freedman GM, Lin L. Cardiac-sparing radiation therapy for breast cancer. Appl Rad Oncol. 2016;(3):6-11.
September 12, 2016